0001034842--12-31falseQ1RIGEL PHARMACEUTICALS INC2026xbrli:sharesiso4217:USDiso4217:USDxbrli:sharesxbrli:purerigl:securityrigl:segment00010348422026-01-012026-03-3100010348422026-04-3000010348422026-03-3100010348422025-12-310001034842us-gaap:ProductMember2026-01-012026-03-310001034842us-gaap:ProductMember2025-01-012025-03-310001034842rigl:ContractRevenuesFromCollaborationsMember2026-01-012026-03-310001034842rigl:ContractRevenuesFromCollaborationsMember2025-01-012025-03-3100010348422025-01-012025-03-310001034842us-gaap:CommonStockMember2025-12-310001034842us-gaap:AdditionalPaidInCapitalMember2025-12-310001034842us-gaap:AccumulatedOtherComprehensiveIncomeMember2025-12-310001034842us-gaap:RetainedEarningsMember2025-12-310001034842us-gaap:RetainedEarningsMember2026-01-012026-03-310001034842us-gaap:AccumulatedOtherComprehensiveIncomeMember2026-01-012026-03-310001034842us-gaap:CommonStockMember2026-01-012026-03-310001034842us-gaap:AdditionalPaidInCapitalMember2026-01-012026-03-310001034842us-gaap:CommonStockMember2026-03-310001034842us-gaap:AdditionalPaidInCapitalMember2026-03-310001034842us-gaap:AccumulatedOtherComprehensiveIncomeMember2026-03-310001034842us-gaap:RetainedEarningsMember2026-03-310001034842us-gaap:CommonStockMember2024-12-310001034842us-gaap:AdditionalPaidInCapitalMember2024-12-310001034842us-gaap:AccumulatedOtherComprehensiveIncomeMember2024-12-310001034842us-gaap:RetainedEarningsMember2024-12-3100010348422024-12-310001034842us-gaap:RetainedEarningsMember2025-01-012025-03-310001034842us-gaap:AccumulatedOtherComprehensiveIncomeMember2025-01-012025-03-310001034842us-gaap:CommonStockMember2025-01-012025-03-310001034842us-gaap:AdditionalPaidInCapitalMember2025-01-012025-03-310001034842us-gaap:CommonStockMember2025-03-310001034842us-gaap:AdditionalPaidInCapitalMember2025-03-310001034842us-gaap:AccumulatedOtherComprehensiveIncomeMember2025-03-310001034842us-gaap:RetainedEarningsMember2025-03-3100010348422025-03-310001034842us-gaap:EmployeeStockOptionMember2026-01-012026-03-310001034842us-gaap:EmployeeStockOptionMember2025-01-012025-03-310001034842us-gaap:RestrictedStockUnitsRSUMember2026-01-012026-03-310001034842us-gaap:RestrictedStockUnitsRSUMember2025-01-012025-03-310001034842us-gaap:EmployeeStockMember2026-01-012026-03-310001034842us-gaap:EmployeeStockMember2025-01-012025-03-310001034842rigl:GrossProductMember2026-01-012026-03-310001034842rigl:GrossProductMember2025-01-012025-03-310001034842rigl:DiscountsAndAllowancesMember2026-01-012026-03-310001034842rigl:DiscountsAndAllowancesMember2025-01-012025-03-310001034842rigl:DevelopmentMilestonesMember2026-01-012026-03-310001034842rigl:DevelopmentMilestonesMember2025-01-012025-03-310001034842rigl:RoyaltyDeliveryOfDrugSuppliesAndOthersMember2026-01-012026-03-310001034842rigl:RoyaltyDeliveryOfDrugSuppliesAndOthersMember2025-01-012025-03-310001034842us-gaap:AllowanceForCreditLossMember2026-01-012026-03-310001034842us-gaap:AllowanceForCreditLossMember2025-01-012025-03-310001034842rigl:ChargebacksDiscountsAndFeesMember2025-12-310001034842rigl:GovernmentAndOtherRebatesMember2025-12-310001034842us-gaap:SalesReturnsAndAllowancesMember2025-12-310001034842rigl:ChargebacksDiscountsAndFeesMember2026-01-012026-03-310001034842rigl:GovernmentAndOtherRebatesMember2026-01-012026-03-310001034842us-gaap:SalesReturnsAndAllowancesMember2026-01-012026-03-310001034842rigl:ChargebacksDiscountsAndFeesMember2026-03-310001034842rigl:GovernmentAndOtherRebatesMember2026-03-310001034842us-gaap:SalesReturnsAndAllowancesMember2026-03-310001034842rigl:ChargebacksDiscountsAndFeesMember2024-12-310001034842rigl:GovernmentAndOtherRebatesMember2024-12-310001034842us-gaap:SalesReturnsAndAllowancesMember2024-12-310001034842rigl:ChargebacksDiscountsAndFeesMember2025-01-012025-03-310001034842rigl:GovernmentAndOtherRebatesMember2025-01-012025-03-310001034842us-gaap:SalesReturnsAndAllowancesMember2025-01-012025-03-310001034842rigl:ChargebacksDiscountsAndFeesMember2025-03-310001034842rigl:GovernmentAndOtherRebatesMember2025-03-310001034842us-gaap:SalesReturnsAndAllowancesMember2025-03-310001034842rigl:MckessonSpecialtyCareDistributionCorporationMemberus-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMember2026-01-012026-03-310001034842rigl:MckessonSpecialtyCareDistributionCorporationMemberus-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMember2025-01-012025-03-310001034842rigl:CencoraIncMemberus-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMember2026-01-012026-03-310001034842rigl:CencoraIncMemberus-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMember2025-01-012025-03-310001034842rigl:OptimeCareIncMemberus-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMember2026-01-012026-03-310001034842rigl:OptimeCareIncMemberus-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMember2025-01-012025-03-310001034842rigl:CardinalHealthIncMemberus-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMember2026-01-012026-03-310001034842us-gaap:CollaborativeArrangementMember2026-01-012026-03-310001034842us-gaap:CollaborativeArrangementMemberrigl:SpecifiedDevelopmentEventsMember2026-01-012026-03-310001034842us-gaap:CollaborativeArrangementMemberrigl:SpecifiedProductLaunchEventsMember2026-01-012026-03-3100010348422021-04-012021-04-300001034842rigl:NonCentralNervousSystemSpecifiedDevelopmentAndRegulatoryMilestoneMember2026-01-012026-03-310001034842rigl:ProductByProductSalesMilestoneMember2026-01-012026-03-310001034842rigl:CentralNervousSystemSpecifiedDevelopmentAndRegulatoryMilestoneMember2026-01-012026-03-310001034842rigl:CentralNervousSystemProductByProductSalesMilestoneMember2026-01-012026-03-310001034842rigl:OcadusertibMember2026-01-012026-03-310001034842rigl:OcadusertibMember2025-04-012025-06-300001034842rigl:OcadusertibMember2026-03-310001034842rigl:OcadusertibMember2025-12-310001034842rigl:GrifolsS.a.Member2026-03-310001034842rigl:GrifolsS.a.Member2025-12-310001034842rigl:GrifolsS.a.Memberrigl:OneTimeDeliveryOfDrugSupplyForCommercializationMember2026-01-012026-03-310001034842rigl:GrifolsS.a.Memberrigl:OneTimeDeliveryOfDrugSupplyForCommercializationMember2025-01-012025-03-310001034842rigl:GrifolsS.a.Memberus-gaap:RoyaltyMember2026-01-012026-03-310001034842rigl:GrifolsS.a.Memberus-gaap:RoyaltyMember2025-01-012025-03-310001034842rigl:KisseiPharmaceuticalCo.Ltd.Member2026-03-310001034842rigl:KisseiPharmaceuticalCo.Ltd.Member2025-12-310001034842rigl:KisseiPharmaceuticalCo.Ltd.Memberrigl:FostamatinibMember2026-03-310001034842rigl:KisseiPharmaceuticalCo.Ltd.Memberrigl:FostamatinibMember2025-12-310001034842rigl:KisseiPharmaceuticalCo.Ltd.Memberrigl:FostamatinibMember2025-03-310001034842rigl:KisseiPharmaceuticalCo.Ltd.Member2026-01-012026-03-310001034842rigl:KisseiPharmaceuticalCo.Ltd.Member2025-01-012025-03-310001034842rigl:MedisonPharmaLtd.Memberrigl:FostamatinibMember2026-03-310001034842rigl:MedisonPharmaLtd.Memberrigl:FostamatinibMember2025-12-310001034842rigl:MedisonPharmaLtd.Memberrigl:FostamatinibMember2026-01-012026-03-310001034842rigl:MedisonPharmaLtd.Memberrigl:FostamatinibMember2025-01-012025-03-310001034842rigl:RezlidhiaMemberrigl:StrategicCollaborationAgreementWithMdaccMember2023-12-310001034842rigl:RezlidhiaMemberrigl:StrategicCollaborationAgreementWithMdaccMember2026-01-012026-03-310001034842rigl:RezlidhiaMemberrigl:StrategicCollaborationAgreementWithMdaccMember2023-12-012026-03-310001034842rigl:RezlidhiaMemberrigl:StrategicCollaborationAgreementWithConnectMember2024-01-310001034842rigl:RezlidhiaMemberrigl:StrategicCollaborationAgreementWithConnectMember2026-01-012026-03-310001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMember2024-02-222024-02-220001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMemberrigl:FirstCommercialSaleMember2024-02-222024-02-220001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMemberrigl:FirstAnniversaryOfClosingDateMember2024-02-222024-02-220001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMemberrigl:SpecifiedCommercialMilestonesMember2024-02-220001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMemberrigl:RegulatoryMilestonesMember2024-02-220001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMembersrt:MinimumMember2024-02-222024-02-220001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMembersrt:MaximumMember2024-02-222024-02-220001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMember2024-07-012024-07-310001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMember2026-03-310001034842rigl:GavretoMemberrigl:BlueprintMedicinesCorporationMember2025-12-310001034842rigl:GavretoMember2026-01-012026-03-310001034842rigl:FormaTherapeuticsHoldingsIncMember2022-07-012022-07-310001034842rigl:FormaTherapeuticsHoldingsIncMemberrigl:DevelopmentAndRegulatoryMilestonesMember2022-07-012022-07-310001034842rigl:FormaTherapeuticsHoldingsIncMemberrigl:SpecifiedCommercialMilestonesMember2022-07-012022-07-310001034842rigl:FormaTherapeuticsHoldingsIncMember2022-01-012022-12-310001034842rigl:FormaTherapeuticsHoldingsIncMemberrigl:AchievementOfCertainNearTermRegulatoryMilestoneMember2022-12-012022-12-310001034842rigl:RezlidhiaMemberrigl:FDAApprovalAndFirstCommercialSaleOfProductMember2022-12-310001034842rigl:RezlidhiaMember2026-01-012026-03-310001034842us-gaap:SellingGeneralAndAdministrativeExpensesMember2026-01-012026-03-310001034842us-gaap:SellingGeneralAndAdministrativeExpensesMember2025-01-012025-03-310001034842us-gaap:ResearchAndDevelopmentExpenseMember2026-01-012026-03-310001034842us-gaap:ResearchAndDevelopmentExpenseMember2025-01-012025-03-310001034842us-gaap:EmployeeStockOptionMember2026-01-012026-03-310001034842us-gaap:RestrictedStockUnitsRSUMember2026-01-012026-03-310001034842us-gaap:EmployeeStockOptionMember2025-01-012025-03-310001034842us-gaap:EmployeeStockOptionMember2026-03-310001034842us-gaap:RestrictedStockUnitsRSUMember2026-03-310001034842us-gaap:EmployeeStockOptionMemberus-gaap:ShareBasedCompensationAwardTrancheOneMember2026-03-310001034842us-gaap:RestrictedStockUnitsRSUMemberus-gaap:ShareBasedCompensationAwardTrancheOneMember2026-03-310001034842rigl:ShareBasedCompensationOptionsAndRsusMember2026-03-310001034842rigl:ShareBasedCompensationOptionsAndRsusMember2026-01-012026-03-310001034842us-gaap:EmployeeStockMember2024-07-012024-07-010001034842us-gaap:EmployeeStockMember2026-03-310001034842us-gaap:EmployeeStockMember2026-01-012026-03-310001034842us-gaap:CashMember2026-03-310001034842us-gaap:CashMember2025-12-310001034842rigl:RestrictedCashMember2026-03-310001034842rigl:RestrictedCashMember2025-12-310001034842us-gaap:MoneyMarketFundsMember2026-03-310001034842us-gaap:MoneyMarketFundsMember2025-12-310001034842us-gaap:USTreasuryBillSecuritiesMember2026-03-310001034842us-gaap:USTreasuryBillSecuritiesMember2025-12-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMember2026-03-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMember2025-12-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMember2026-03-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMember2025-12-310001034842us-gaap:USTreasuryBillSecuritiesMember2026-03-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMember2026-03-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMember2026-03-310001034842us-gaap:USTreasuryBillSecuritiesMember2025-12-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMember2025-12-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMember2025-12-3100010348422025-01-012025-12-310001034842us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2026-03-310001034842us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel2Member2026-03-310001034842us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2026-03-310001034842us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueInputsLevel1Member2026-03-310001034842us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueInputsLevel2Member2026-03-310001034842us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueInputsLevel3Member2026-03-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMemberus-gaap:FairValueInputsLevel1Member2026-03-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMemberus-gaap:FairValueInputsLevel2Member2026-03-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMemberus-gaap:FairValueInputsLevel3Member2026-03-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMemberus-gaap:FairValueInputsLevel1Member2026-03-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMemberus-gaap:FairValueInputsLevel2Member2026-03-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMemberus-gaap:FairValueInputsLevel3Member2026-03-310001034842us-gaap:FairValueInputsLevel1Member2026-03-310001034842us-gaap:FairValueInputsLevel2Member2026-03-310001034842us-gaap:FairValueInputsLevel3Member2026-03-310001034842us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel1Member2025-12-310001034842us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel2Member2025-12-310001034842us-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2025-12-310001034842us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueInputsLevel1Member2025-12-310001034842us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueInputsLevel2Member2025-12-310001034842us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueInputsLevel3Member2025-12-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMemberus-gaap:FairValueInputsLevel1Member2025-12-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMemberus-gaap:FairValueInputsLevel2Member2025-12-310001034842us-gaap:USGovernmentSponsoredEnterprisesDebtSecuritiesMemberus-gaap:FairValueInputsLevel3Member2025-12-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMemberus-gaap:FairValueInputsLevel1Member2025-12-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMemberus-gaap:FairValueInputsLevel2Member2025-12-310001034842rigl:CorporateBondSecuritiesAndCommercialPaperMemberus-gaap:FairValueInputsLevel3Member2025-12-310001034842us-gaap:FairValueInputsLevel1Member2025-12-310001034842us-gaap:FairValueInputsLevel2Member2025-12-310001034842us-gaap:FairValueInputsLevel3Member2025-12-310001034842us-gaap:LineOfCreditMember2026-03-310001034842us-gaap:LineOfCreditMemberrigl:OneMonthSecuredOvernightFinancingRateSOFRMember2026-01-012026-03-310001034842us-gaap:LineOfCreditMemberus-gaap:BaseRateMember2026-01-012026-03-310001034842us-gaap:LineOfCreditMemberus-gaap:SecuredOvernightFinancingRateSofrMember2026-01-012026-03-310001034842us-gaap:LineOfCreditMember2026-01-012026-03-310001034842rigl:HeadquartersOfficeSpaceSubleaseSouthSanFranciscoCaliforniaMember2026-03-310001034842country:US2026-01-012026-03-310001034842us-gaap:StateAndLocalTaxJurisdictionOtherMember2026-01-012026-03-310001034842rigl:OneOperatingSegmentMember2026-01-012026-03-310001034842rigl:OneOperatingSegmentMember2025-01-012025-03-310001034842us-gaap:LineOfCreditMemberus-gaap:SubsequentEventMember2026-05-040001034842us-gaap:LineOfCreditMemberus-gaap:SubsequentEventMember2026-05-050001034842us-gaap:LineOfCreditMemberus-gaap:BaseRateMemberus-gaap:SubsequentEventMember2026-05-050001034842us-gaap:LineOfCreditMemberus-gaap:SecuredOvernightFinancingRateSofrMemberus-gaap:SubsequentEventMember2026-05-052026-05-05
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
_______________________________________________________________
FORM 10-Q
(Mark One)
| | | | | |
| x | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
FOR THE QUARTERLY PERIOD ENDED MARCH 31, 2026
OR
| | | | | |
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
FOR THE TRANSITION PERIOD FROM TO
Commission File Number 0-29889
_______________________________________________________________
Rigel Pharmaceuticals, Inc.
(Exact name of registrant as specified in its charter)
| | | | | |
| Delaware | 94-3248524 |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
| |
611 Gateway Boulevard, Suite 900, | |
South San Francisco, CA | 94080 |
| (Address of principal executive offices) | (Zip Code) |
(650) 624-1100
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | | | | | | | |
| Title of each class: | | Trading Symbol | | Name of each exchange on which registered: |
| Common Stock, par value $0.001 per share | | RIGL | | The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No o
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes x No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | |
| Large accelerated filer | o | Accelerated filer | x |
| Non-accelerated filer | o | Smaller reporting company | o |
| Emerging Growth Company | o | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒
As of April 30, 2026, there were 18,502,080 shares of the registrant’s Common Stock outstanding.
RIGEL PHARMACEUTICALS, INC.
QUARTERLY REPORT ON FORM 10-Q
FOR THE QUARTERLY PERIOD ENDED MARCH 31, 2026
INDEX
PART I. FINANCIAL INFORMATION
Item 1. Financial Statements
RIGEL PHARMACEUTICALS, INC.
CONDENSED BALANCE SHEETS
(In thousands)
| | | | | | | | | | | |
| As of |
| March 31, 2026 | | December 31, 2025 ⁽¹⁾ |
| (unaudited) | | |
| Assets | | | |
| Current assets: | | | |
| Cash and cash equivalents | $ | 24,411 | | | $ | 40,580 | |
| Short-term investments | 122,273 | | | 114,375 | |
| Accounts receivable, net | 49,945 | | | 51,763 | |
| Inventories | 11,652 | | | 11,506 | |
| Prepaid and other current assets | 25,746 | | | 21,942 | |
| Total current assets | 234,027 | | | 240,166 | |
| | | |
| Intangible assets, net | 24,160 | | | 24,748 | |
| Deferred income tax asset | 243,279 | | | 245,852 | |
| Operating lease right-of-use assets | 785 | | | 920 | |
| Other assets | 2,357 | | | 1,908 | |
| Total assets | $ | 504,608 | | | $ | 513,594 | |
| Liabilities and stockholders’ equity | | | |
| Current liabilities: | | | |
| Accounts payable | $ | 4,486 | | | $ | 7,191 | |
| Accrued compensation | 6,009 | | | 11,914 | |
| Accrued research and development | 5,742 | | | 5,524 | |
| Acquisition-related liabilities | 5,000 | | | 5,000 | |
| Revenue reserves and refund liability | 28,080 | | | 27,716 | |
| Loans payable, net, current portion | 29,865 | | | 29,812 | |
| Other accrued liabilities | 9,674 | | | 11,466 | |
| | | |
| Lease liabilities, current portion | 636 | | | 614 | |
| Total current liabilities | 89,492 | | | 99,237 | |
| | | |
| Long-term portion of lease liabilities | 228 | | | 395 | |
| Long-term portion of loans payable, net | 14,991 | | | 22,482 | |
| | | |
| Total liabilities | 104,711 | | | 122,114 | |
| | | |
| Commitments | | | |
| | | |
| Stockholders’ equity: | | | |
| Common stock | 18 | | | 18 | |
| Additional paid-in capital | 1,414,394 | | | 1,414,322 | |
| Accumulated other comprehensive (loss) income | (128) | | | 181 | |
| Accumulated deficit | (1,014,387) | | | (1,023,041) | |
| Total stockholders’ equity | 399,897 | | | 391,480 | |
| Total liabilities and stockholders’ equity | $ | 504,608 | | | $ | 513,594 | |
______________________________________________________________________
(1)The balance sheet as of December 31, 2025 has been derived from the audited financial statements included in Rigel’s Annual Report on Form 10-K for the year ended December 31, 2025 filed with the Securities and Exchange Commission (SEC) on March 3, 2026.
See Accompanying Notes to Condensed Financial Statements
RIGEL PHARMACEUTICALS, INC.
CONDENSED STATEMENTS OF OPERATIONS
(In thousands, except per share amounts)
(unaudited)
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| Revenues: | | | | | | | |
| Product sales, net | $ | 54,923 | | | $ | 43,550 | | | | | |
| Contract revenues from collaborations | 3,895 | | | 9,783 | | | | | |
| | | | | | | |
| Total revenues | 58,818 | | | 53,333 | | | | | |
| Costs and expenses: | | | | | | | |
| Cost of product sales | 4,606 | | | 4,409 | | | | | |
| Research and development | 11,676 | | | 8,436 | | | | | |
| Selling, general and administrative | 30,651 | | | 27,715 | | | | | |
| Total costs and expenses | 46,933 | | | 40,560 | | | | | |
| Income from operations | 11,885 | | | 12,773 | | | | | |
| Interest income | 1,205 | | | 591 | | | | | |
| Interest expense | (1,433) | | | (1,853) | | | | | |
| Income before income taxes | 11,657 | | | 11,511 | | | | | |
| Provision for income taxes | 3,003 | | | 65 | | | | | |
| Net income | $ | 8,654 | | | $ | 11,446 | | | | | |
| | | | | | | |
| Net income per share | | | | | | | |
| Basic | $ | 0.47 | | | $ | 0.64 | | | | | |
| Diluted | $ | 0.44 | | | $ | 0.63 | | | | | |
| Weighted average shares used in computing net income per share | | | | | | | |
| Basic | 18,412 | | 17,808 | | | | |
| Diluted | 19,686 | | 18,169 | | | | |
See Accompanying Notes to Condensed Financial Statements
RIGEL PHARMACEUTICALS, INC.
CONDENSED STATEMENTS OF COMPREHENSIVE INCOME
(In thousands)
(unaudited)
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| Net income | $ | 8,654 | | | $ | 11,446 | | | | | |
| Other comprehensive loss: | | | | | | | |
| Net unrealized loss on short-term investments | (309) | | | (12) | | | | | |
| Comprehensive income | $ | 8,345 | | | $ | 11,434 | | | | | |
See Accompanying Notes to Condensed Financial Statements
RIGEL PHARMACEUTICALS, INC.
CONDENSED STATEMENTS OF STOCKHOLDERS’ EQUITY
(In thousands, except share amounts)
(unaudited)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Common Stock | | Additional
Paid-in
Capital | | Accumulated Other
Comprehensive
Income (Loss) | | Accumulated
Deficit | | Total
Stockholders’
Equity |
| Shares | | Amount | | | | |
| Balance as of January 1, 2026 | 18,310,934 | | $ | 18 | | | $ | 1,414,322 | | | $ | 181 | | | $ | (1,023,041) | | | $ | 391,480 | |
| Net income | — | | — | | | — | | | — | | | 8,654 | | | 8,654 | |
| Net change in unrealized loss on short-term investments | — | | — | | | — | | | (309) | | | — | | | (309) | |
| Issuance of common stock upon exercise of options, net of shares withheld | 50,157 | | — | | | 321 | | | — | | | — | | | 321 | |
| Issuance of common stock upon vesting of restricted stock units (RSUs) | 226,221 | | — | | | — | | | — | | | — | | | — | |
| Repurchases of common stock in connection with employee tax withholding on RSU vesting | (105,728) | | — | | | (3,740) | | | — | | | — | | | (3,740) | |
| Stock-based compensation expense | — | | — | | | 3,491 | | | — | | | — | | | 3,491 | |
| Balance as of March 31, 2026 | 18,481,584 | | $ | 18 | | | $ | 1,414,394 | | | $ | (128) | | | $ | (1,014,387) | | | $ | 399,897 | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Common Stock | | Additional
Paid-in
Capital | | Accumulated Other
Comprehensive
Income (Loss) | | Accumulated
Deficit | | Total
Stockholders’
Equity |
| Shares | | Amount | | | | |
| Balance as of January 1, 2025 | 17,710,216 | | $ | 18 | | | $ | 1,393,325 | | | $ | 10 | | | $ | (1,390,065) | | | $ | 3,288 | |
| Net income | — | | — | | | — | | | — | | | 11,446 | | | 11,446 | |
| Net change in unrealized loss on short-term investments | — | | — | | | — | | | (12) | | | — | | | (12) | |
| Issuance of common stock upon exercise of options, net of shares withheld | 30,892 | | — | | | 484 | | | — | | | — | | | 484 | |
| Issuance of common stock upon vesting of RSUs | 125,783 | | — | | | — | | | — | | | — | | | — | |
| Stock-based compensation expense | — | | — | | | 3,361 | | | — | | | — | | | 3,361 | |
| Balance as of March 31, 2025 | 17,866,891 | | $ | 18 | | | $ | 1,397,170 | | | $ | (2) | | | $ | (1,378,619) | | | $ | 18,567 | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
See Accompanying Notes to Condensed Financial Statements
RIGEL PHARMACEUTICALS, INC.
CONDENSED STATEMENTS OF CASH FLOWS
(In thousands)
(unaudited)
| | | | | | | | | | | |
| Three Months Ended March 31, |
| 2026 | | 2025 |
| Operating activities | | | |
| Net income | $ | 8,654 | | | $ | 11,446 | |
| Adjustments to reconcile net income to net cash provided by (used in) operating activities: | | | |
| Stock-based compensation expense | 3,456 | | | 3,324 | |
| | | |
| Depreciation and amortization | 597 | | | 604 | |
| Deferred income tax | 2,573 | | | — | |
| | | |
| Net amortization of discount on short-term investments and term loans | (170) | | | (190) | |
| Changes in assets and liabilities: | | | |
| Accounts receivable, net | 1,818 | | | 68 | |
| Inventories | (581) | | | 1,413 | |
| Prepaid and other current and non-current assets | (3,792) | | | (13,150) | |
| Right-of-use assets | 135 | | | (1,059) | |
| Accounts payable | (2,705) | | | 615 | |
| Accrued compensation | (5,905) | | | (4,115) | |
| Accrued research and development | 218 | | | 575 | |
| Revenue reserves and refund liability | 364 | | | 1,495 | |
| Other accrued liabilities | (1,792) | | | (2,954) | |
| Lease liabilities | (145) | | | 1,035 | |
| Net cash provided by (used in) operating activities | 2,725 | | | (893) | |
| Investing activities | | | |
| Maturities of short-term investments | 15,945 | | | 11,100 | |
| Purchases of short-term investments | (23,920) | | | (21,652) | |
| | | |
| | | |
| | | |
| Net cash used in investing activities | (7,975) | | | (10,552) | |
| Financing activities | | | |
| | | |
| Principal payments of term loans | (7,500) | | | — | |
| Net proceeds from issuance of common stock under equity plans | 321 | | | 484 | |
| Repurchases of common stock in connection with employee tax withholding on RSU vesting | (3,740) | | | — | |
| | | |
| Net cash (used in) provided by financing activities | (10,919) | | | 484 | |
| Net decrease in cash, cash equivalents and restricted cash | (16,169) | | | (10,961) | |
| Cash, cash equivalents, and restricted cash at beginning of period | 40,637 | | | 56,746 | |
| Cash, cash equivalents, and restricted cash at end of period | $ | 24,468 | | | $ | 45,785 | |
| Supplemental disclosure of cash flow information | | | |
| Interest paid | $ | 1,320 | | | $ | 1,653 | |
| Increase in right-of-use assets and lease liabilities | $ | — | | | $ | 1,220 | |
| Acquisition-related liabilities | $ | 5,000 | | | $ | 5,000 | |
See Accompanying Notes to Condensed Financial Statements
Rigel Pharmaceuticals, Inc.
Notes to Condensed Financial Statements
(unaudited)
In this report, “Rigel,” “we,” “us” and “our” refer to Rigel Pharmaceuticals, Inc.
1. Organization and Summary of Significant Accounting Policies
Description of Business
We are a biotechnology company dedicated to developing and providing novel therapies that significantly improve the lives of patients with hematologic disorders and cancer. We focus on products that address signaling pathways that are critical to disease mechanisms.
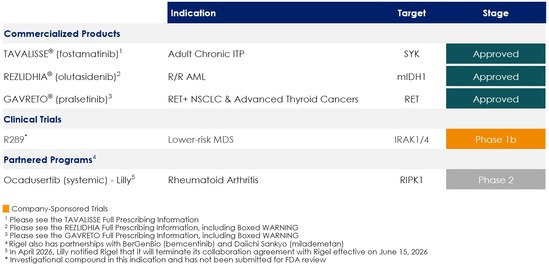
TAVALISSE® (fostamatinib disodium hexahydrate) is our first US Food and Drug Administration (FDA)-approved product and is the only approved oral spleen tyrosine kinase (SYK) inhibitor for the treatment of adult patients with chronic immune thrombocytopenia (ITP) who have had an insufficient response to a previous treatment. The product is also commercially available in Europe and the United Kingdom (UK) (as TAVLESSE), and in Japan, the Republic of Korea (Korea), Canada and Israel (as TAVALISSE) for the treatment of chronic ITP in adult patients.
REZLIDHIA® (olutasidenib) is our second FDA-approved product and indicated for the treatment of adult patients with relapsed or refractory (R/R) acute myeloid leukemia (AML) with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation as detected by an FDA-approved test. We in-licensed REZLIDHIA from Forma Therapeutics, Inc., now Novo Nordisk (Forma), with exclusive, worldwide rights for its development, manufacturing and commercialization, pursuant to a license and transition services agreement entered in July 2022.
GAVRETO® (pralsetinib) is our third FDA-approved product which we began commercializing in June 2024. GAVRETO is a once daily, small molecule, oral, kinase inhibitor of wild-type rearranged during transfection (RET) and oncogenic RET fusions. GAVRETO is approved by the FDA for the treatment of adult patients with metastatic RET fusion-positive non-small cell lung cancer (NSCLC) as detected by an FDA-approved test. GAVRETO is also approved under accelerated approval based on overall response rate and duration response rate, for the treatment of adult and pediatric patients 12 years of age and older with advanced or metastatic RET fusion-positive thyroid cancer who require systemic therapy and who are radioactive iodine-refractory (if radioactive iodine is appropriate). We acquired the rights to research, develop, manufacture and commercialize GAVRETO in the US from Blueprint Medicines Corporation, now a Sanofi SA company (Blueprint), pursuant to an asset purchase agreement entered in February 2024.
Our development pipeline includes R289, our dual interleukin receptor-associated kinases 1 and 4 (IRAK1/4) inhibitor program, which is being advanced in an open-label, Phase 1b study to determine the safety, tolerability and preliminary efficacy of the drug in patients with lower-risk myelodysplastic syndrome (MDS) who are relapsed, refractory or resistant to prior therapies.
To expand our evaluation of olutasidenib in other disease areas with IDH1 mutations, we have strategic development collaborations with the University of Texas MD Anderson Cancer Center (MDACC) and with Collaborative Network for Neuro-Oncology Clinical Trials (CONNECT).
Basis of Presentation
Our accompanying unaudited condensed financial statements have been prepared in accordance with United States generally accepted accounting principles (US GAAP), for interim financial information and pursuant to the instructions to Form 10-Q and Article 10 of Regulation S-X of the Securities Act of 1933, as amended (Securities Act). Accordingly, they do not include all the information and notes required by US GAAP for complete financial statements.
These unaudited condensed financial statements include only normal and recurring adjustments that we believe are necessary to fairly state our financial position and the results of our operations and cash flows. Interim-period results are not necessarily indicative of results of operations or cash flows for a full-year or any subsequent interim period. The balance sheet as of December 31, 2025 has been derived from audited financial statements at that date but does not include all disclosures required by US GAAP for complete financial statements. Because certain disclosures required by US GAAP for complete financial statements are not included herein, these interim unaudited condensed financial statements and the notes accompanying them should be read in conjunction with our audited financial statements and the notes thereto included in our Annual Report on Form 10-K for the year ended December 31, 2025.
Use of Estimates
The preparation of the accompanying unaudited condensed financial statements in conformity with US GAAP requires management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances. Actual results could differ from these estimates.
Significant Accounting Policies
Our significant accounting policies are described in “Note 1 – Description of Business and Summary of Significant Accounting Policies” to our “Notes to Financial Statements” contained in Part II, Item 8, “Financial Statements and Supplementary Data” of our Annual Report on Form 10-K for the year ended December 31, 2025.
Liquidity
As of March 31, 2026, we had approximately $146.7 million in cash, cash equivalents and short-term investments. We finance our operations primarily through sales of our products, and contract payments under our collaboration agreements, as well as through equity securities and debt financing.
Based on our current operating plan, we believe that our existing cash, cash equivalents, and short-term investments will be sufficient to fund our expenses and capital expenditure requirements for at least the next 12 months from the date of issuance of this Form 10-Q.
Recently Issued Accounting Standards
In November 2024, the FASB issued ASU 2024-03, Income Statement—Reporting Comprehensive Income—Expense Disaggregation Disclosures. This guidance enhances expense disclosure requirements for a public business entity’s expenses by mandating detailed information regarding types of expenses (including purchases of inventory, employee compensation, depreciation and amortization) contained in income statement expense categories. This guidance is effective for our annual reporting for the fiscal year ending December 31, 2027, and interim reporting periods starting with the fiscal year ending December 31, 2028, though early adoption is available. This guidance may be applied prospectively to reporting periods after the effective date or retrospectively to all periods presented in the financial statements. We are currently evaluating this guidance and assessing the potential impact on our financial statements and disclosures.
In July 2025, the FASB issued ASU 2025-05, Measurement of Credit Losses for Accounts Receivable and Contract Assets, introducing a practical expedient for credit loss measurement on accounts receivable and contract assets. This guidance is effective for our annual reporting period for the fiscal year ending December 31, 2026, including interim periods within such reporting period, though early adoption is available. The adoption of this guidance did not have a material impact on our financial statements and disclosures.
In December 2025, the FASB issued ASU 2025-10, Accounting for Government Grants Received by Business Entities, which establishes the accounting and presentation for government grants received by a business entity. This guidance is effective for our annual reporting period for the fiscal year ending December 31, 2029, and related interim periods, though early adoption is available. Organizations may adopt this guidance using modified prospective, modified retrospective, or retrospective approaches. We are currently evaluating this guidance and assessing the potential impact on our financial statements and disclosures.
In December 2025, the FASB issued ASU 2025-11, Interim Reporting (Topic 270): Narrow-Scope Improvements to improve the guidance in Topic 270, Interim Reporting. The update provides clarifications aimed at improving interim disclosure requirement consistency and usability, incorporating a comprehensive listing of required interim disclosures and a new disclosure principle for reporting material events occurring after the most recent annual period. The amendments do not change the underlying objectives of interim reporting but are designed to enhance clarity in application. This guidance is effective for our interim reporting periods for the fiscal year ending December 31, 2028. We are currently evaluating the impact of adoption of this standard on our financial statements and disclosures.
In December 2025, the FASB issued ASU 2025-12, Codification Improvements, which addresses thirty-three issues, representing amendments to Accounting Standard Codification topics that clarify, correct errors or make minor improvements. The amendments make the Codification easier to understand and apply. The update is effective for us in our annual reporting period for the fiscal year ending December 31, 2027, and interim periods within such reporting period.
Early adoption and retrospective application are permitted on an issue-by-issue basis. We are currently evaluating this guidance and assessing the potential impact on our financial statements and disclosures.
Other recently issued accounting guidance not discussed in this Quarterly Report on Form 10-Q are either not applicable or did not have, or are not expected to have, a material impact on us.
2. Net Income Per Share
The following table sets forth the computation of basic and diluted earnings per share (in thousands except per share amounts):
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| EPS Numerator: | | | | | | | |
| Net income | $ | 8,654 | | | $ | 11,446 | | | | | |
| EPS Denominator—Basic: | | | | | | | |
| Weighted-average common shares outstanding | 18,412 | | 17,808 | | | | |
| EPS Denominator—Diluted: | | | | | | | |
| Weighted-average common shares outstanding | 18,412 | | 17,808 | | | | |
| Dilutive effect of stock options, RSUs and shares under Purchase Plan | 1,274 | | 361 | | | | |
| Weighted-average shares outstanding and common stock equivalents | 19,686 | | 18,169 | | | | |
| | | | | | | |
| Net income per share | | | | | | | |
| Basic | $ | 0.47 | | | $ | 0.64 | | | | | |
| Diluted | $ | 0.44 | | | $ | 0.63 | | | | | |
The potential shares of common stock that were excluded from the computation of diluted net income per share for the periods presented because including them would have been antidilutive are as follows (in thousands):
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| Stock options | 936 | | | 2,582 | | | | | |
| RSUs | 416 | | | 231 | | | | | |
| Shares under Purchase Plan | — | | | 25 | | | | | |
| Total | 1,352 | | | 2,838 | | | | | |
3. Revenues
Revenues disaggregated by category were as follows (in thousands):
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| Product sales: | | | | | | | |
| Gross product sales | $ | 75,388 | | | $ | 60,192 | | | | | |
| Discounts and allowances | (20,465) | | | (16,642) | | | | | |
| Total product sales, net | 54,923 | | | 43,550 | | | | | |
| Contract revenues from collaborations: | | | | | | | |
| | | | | | | |
| | | | | | | |
| Milestone revenue | — | | | 3,000 | | | | | |
| Delivery of drug supplies, royalties and others | 3,895 | | | 6,783 | | | | | |
| Total contract revenues from collaborations | 3,895 | | | 9,783 | | | | | |
| | | | | | | |
| Total revenues | $ | 58,818 | | | $ | 53,333 | | | | | |
Revenue from product sales relates to sales of our commercial products to customers. For additional information regarding revenue from collaborations, see “Note 4 – Sponsored Research and License Agreements.”
Net product sales represent gross product sales less chargebacks, discounts and fees, government and other rebates, and returns. Of the total discounts and allowances from gross product sales presented in the table above for the three months ended March 31, 2026 and 2025, $18.9 million and $16.1 million, respectively, were accounted for as additions to revenue reserves and refund liability, and $1.5 million and $0.5 million, respectively, were accounted for as reductions to accounts receivable (prompt pay discount) and prepaid and other current assets (for certain prepaid fees) in the condensed balance sheet.
The following tables summarize activity in chargebacks, discounts and fees, government and other rebates, and returns included in revenue reserves and refund liabilities for each of the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Chargebacks,
Discounts and
Fees | | Government
and Other
Rebates | | Returns | | Total |
| Balance as of January 1, 2026 | $ | 12,519 | | | $ | 10,389 | | | $ | 4,808 | | | $ | 27,716 | |
| Provision related to current period sales | 14,379 | | | 4,766 | | | 550 | | | 19,695 | |
| Adjustment related to prior period sales | (363) | | | (381) | | | — | | | (744) | |
| Credit or payments made during the period | (14,459) | | | (3,848) | | | (280) | | | (18,587) | |
| Balance as of March 31, 2026 | $ | 12,076 | | | $ | 10,926 | | | $ | 5,078 | | | $ | 28,080 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| Chargebacks,
Discounts and
Fees | | Government
and Other
Rebates | | Returns | | Total |
| Balance as of January 1, 2025 | $ | 13,374 | | | $ | 8,343 | | | $ | 4,723 | | | $ | 26,440 | |
| Provision related to current period sales | 12,811 | | | 3,654 | | | 398 | | | 16,863 | |
| Adjustment related to prior period sales | — | | | (148) | | | (560) | | | (708) | |
| Credit or payments made during the period | (12,928) | | | (1,669) | | | (63) | | | (14,660) | |
| Balance as of March 31, 2025 | $ | 13,257 | | | $ | 10,180 | | | $ | 4,498 | | | $ | 27,935 | |
Adjustment related to prior period sales reflect updates to estimates of variable consideration, including chargebacks, rebates, and returns, resulting from actual claims and other information obtained in the current reporting period.
The following table summarizes the percentages of revenues from each of our customers who individually accounted for 10% or more of the total net product sales and revenues from collaborations:
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| McKesson Corporation | 48 | % | | 42 | % | | | | |
| Cencora, Inc. | 23 | % | | 20 | % | | | | |
| Optime Care, Inc. | — | | | 11% | | | | |
| Cardinal Health, Inc. | 12 | % | | * | | | | |
| | | | | | | |
| | | | | | | |
______________________________________________________________________
*Denotes less than 10%
4. Sponsored Research and License Agreements
We conduct research and development programs independently and in connection with our corporate collaborators. We were a party to a collaboration agreement with Eli Lilly and Company (Lilly), which included the development and commercialization of ocadusertib (previously R552), a receptor-interacting protein kinase 1 (RIPK1) inhibitor; however, in April 2026, we received a written notice from Lilly to terminate the agreement, as discussed in more detail below.
We are currently a party to collaboration agreements with Grifols S.A. (Grifols) to commercialize fostamatinib for human diseases in all indications in Grifols territory which includes Europe, the UK, Turkey, the Middle East, North Africa and Russia (including Commonwealth of Independent States (CIS)); with Kissei Pharmaceutical Co., Ltd. (Kissei) to develop and commercialize fostamatinib in Japan, China, Taiwan and Korea, and olutasidenib in Japan, Korea and Taiwan; with Medison Pharma Trading AG (Medison Canada) and Medison Pharma Ltd. (Medison Israel and, together with Medison Canada, Medison) to commercialize fostamatinib in all indications, in Medison territory which includes Canada and Israel; with Knight Therapeutics International SA (Knight) to commercialize fostamatinib in all indications, in Knight territory which includes Latin America, consisting of Mexico, Central and South America, and the Caribbean; and with Dr. Reddy’s Laboratories (Dr. Reddy’s) to commercialize olutasidenib in Dr. Reddy’s territory which includes Latin America, South Africa, India, Australia, New Zealand, and certain countries in the CIS, Southeast Asia region and North Africa.
Further, we are also a party to collaboration agreements, but do not have ongoing performance obligations with BerGenBio ASA, now Oncoinvent ASA (BerGenBio) for the development and commercialization of AXL receptor tyrosine kinase inhibitor, R428 (now referred to as bemcentinib (BGB324)), and with Daiichi Sankyo (Daiichi) to pursue research related to murine double minute 2 (MDM2) inhibitor, DS-3032 (now referred as milademetan).
Under the above collaboration agreements, we have received, and may in the future receive, milestone payments contingent upon the achievement of specified events, as well as royalties on net sales of products commercialized by our partners. The total potential future contingent payments under these agreements were approximately $657.1 million. This amount accounts for terminated programs and Lilly’s April 2026 notice to terminate the Lilly Agreement, as discussed in detail below, and assumes the achievement of all applicable milestones under the existing agreements. Of this amount, $195.1 million relates to development and regulatory milestones and $462.0 million to commercial milestones. This estimate excludes any potential royalties that may become payable if our partners successfully commercialize licensed products. Milestone payments under these agreements are contingent upon our partners’ future efforts and the achievement of specified development, regulatory and commercial milestones.
We also have strategic development collaborations with MDACC and CONNECT to expand the evaluation of olutasidenib in other disease areas with IDH1 mutations.
Global Exclusive License Agreement with Lilly
In February 2021, we entered a global exclusive license agreement and strategic collaboration with Lilly, which became effective in March 2021 upon clearance under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, and was amended in September 2023, March 2024, and in August 2025 (collectively, Lilly Agreement). Pursuant to the terms of the Lilly Agreement, we granted Lilly the exclusive rights to develop and commercialize ocadusertib (previously R552) and related receptor interacting serine/threonine protein kinase 1 (RIPK1) inhibitors in all indications worldwide. The
collaboration was to develop and commercialize ocadusertib for the treatment of non-central nervous system (non-CNS) diseases, and additional RIPK1 inhibitors for the treatment of central nervous system (CNS) diseases.
Under the Lilly Agreement, we received a non-refundable and non-creditable upfront cash payment amounting to $125.0 million in April 2021. In addition, for non-CNS diseases, we were eligible to receive up to $330.0 million in development and regulatory milestones and up to $100.0 million in sales milestones on a product-by-product basis, as well as tiered royalties on net sales ranging from the mid-single digits to high-teens, subject to certain standard reductions and offsets. For CNS diseases, we were eligible to receive up to $256.0 million in development, regulatory and commercial milestones and up to $150.0 million in sales milestone payments, as well as tiered royalties on net sales up to low-double digits.
On April 16, 2026, we received a written notice from Lilly of its decision to terminate the Lilly Agreement, which will become effective June 15, 2026. Following termination of the Lilly Agreement, including the prior termination of the CNS disease program effective in November 2025, we do not expect to receive future milestones or royalties under the Lilly Agreement.
Under the Lilly Agreement, we were responsible for 20% of the development costs for ocadusertib in the US, Europe, and Japan, up to a specified cap, and Lilly was responsible for funding the remainder of all development activities for ocadusertib and other non-CNS disease development candidates. In September 2023, we exercised our first opt-out right, and concurrently, our cost share obligation for ocadusertib development ended on April 1, 2024. Although we retained a right to opt back into co-funding to receive increased royalties, we notified Lilly in April 2025 that we would not exercise this option. As a result, we have no further development cost-sharing obligations. In connection with this decision, we released the $40.0 million remaining cost share liability and recognized the amount as contract revenues from collaboration in the second quarter of 2025. As of March 31, 2026 and December 31, 2025, there was no deferred revenue related to the Lilly Agreement.
Grifols License Agreement
We have an exclusive commercialization license agreement with Grifols entered in January 2019 with exclusive rights to commercialize fostamatinib for human diseases, and non-exclusive rights to develop fostamatinib in Grifols territory. There was no deferred revenue related to the Grifols license agreement as of March 31, 2026 and December 31, 2025.
Pursuant to our commercial supply agreement with Grifols, no revenue was recognized from delivery of drug supplies to Grifols for the three months ended March 31, 2026. Revenue recognized for such deliveries was $3.3 million for the three months ended March 31, 2025.
We also recognize royalty revenue from Grifols included within contract revenues from this collaboration. Royalty revenue for the three months ended March 31, 2026 and 2025 from Grifols was $1.8 million and $1.4 million, respectively.
Kissei License Agreements
We have a collaboration and license agreement with Kissei entered in September 2024 to grant exclusive rights to Kissei to develop and commercialize olutasidenib in all human diseases in related Kissei territory. There was no deferred revenue related to this related collaboration and license agreement as of March 31, 2026 and December 31, 2025.
We also have an exclusive license and supply agreement with Kissei entered in October 2018, amended in November 2022, October 2023, August 2024, September 2024 and October 2024, to develop and commercialize fostamatinib in all current and potential indications in related Kissei territory. As of March 31, 2026 and December 31, 2025, the remaining deferred revenue was related to the material right associated with discounted fostamatinib supply which amounted to $1.4 million. During the three months ended March 31, 2025, we recognized $3.0 million of contract revenue from collaborations related to a non-refundable and non-creditable milestone payment from Kissei in connection with the approval of fostamatinib in Korea.
Pursuant to our supply agreement with Kissei, for the three months ended March 31, 2026 and 2025, we recognized $1.8 million and $1.6 million, respectively, of revenue related to delivery of drug supplies to Kissei.
Medison Commercial and License Agreements
We have exclusive commercial and license agreements with Medison entered in October 2019 for the commercialization of fostamatinib for chronic ITP in Medison territory. There was no deferred revenue related to Medison commercial and license agreement as of March 31, 2026 and December 31, 2025.
Revenue recognized from Medison related to delivery of drug supplies and royalties for the three months ended March 31, 2026 and 2025 was $0.3 million and $0.4 million, respectively.
Strategic Development Collaborations with MDACC and CONNECT
We have a Strategic Collaboration Agreement with MDACC entered in December 2023 to evaluate olutasidenib in AML and other hematologic cancers. Under the agreement, we are obligated to provide study materials and up to $15.0 million in time-based milestone payments over the five-year collaboration term, unless terminated earlier. Through March 31, 2026, we have provided $5.3 million in funding to MDACC.
In January 2024, we announced our collaboration with CONNECT to conduct a Phase 2 clinical trial to evaluate olutasidenib in glioma. Under this agreement, we are obligated to provide study materials and up to $3.0 million in funding over the four-year collaboration term.
Amounts paid under these research collaboration agreements are recorded as prepaid research and development to the extent payments are made in advance of services and are recognized as research and development expense as the services are performed.
5. In-licensing and Acquisition
Asset Purchase Agreement with Blueprint
We acquired the US rights to research, develop, manufacture and commercialize GAVRETO from Blueprint pursuant to an Asset Purchase Agreement entered in February 2024. The acquired assets from Blueprint include, among other things, applicable intellectual property related to pralsetinib in the US, including patents, copyrights and trademarks, as well as clinical regulatory and commercial data and records. Pursuant to the Asset Purchase Agreement, we agreed to pay a purchase price of $15.0 million, of which, $10.0 million was payable upon our first commercial sale of GAVRETO and an additional $5.0 million is payable on the first anniversary of the closing date of the agreement, subject to certain conditions. Blueprint is also eligible to receive up to $97.5 million in future commercial milestone payments and up to $5.0 million in future regulatory milestone payments. Blueprint is also entitled to tiered royalty payments on net sales of products containing pralsetinib (or related compounds) ranging from 10% to 30%, subject to certain reductions and offsets.
The total purchase price consideration amounted to $15.4 million, consisting of $15.0 million closing purchase price and $0.4 million of transaction costs. Of the total closing purchase price, $10.0 million was paid in July 2024, and the remaining $5.0 million was outstanding as of March 31, 2026 and December 31, 2025 and presented as acquisition-related liabilities in the condensed balance sheets.
We accounted for this transaction as an asset acquisition in accordance with ASC 805 Business Combinations (ASC 805) and recorded the total purchase consideration as intangible assets at the acquisition date. The intangible assets are amortized on a straight-line basis over an estimated useful life of 12 years, with amortization recognized in cost of product sales. Royalties are also recognized in cost of product sales, as the related product sales occur.
License and Transition Services Agreement with Forma
We have a license and transition services agreement with Forma entered in July 2022, for an exclusive license to develop, manufacture and commercialize olutasidenib, a proprietary inhibitor of mutated IDH1 (mIDH1), for any uses worldwide, including for the treatment of AML and other malignancies. Under the terms of the license and transition services agreement, we paid an upfront fee of $2.0 million, and may be required to pay up to an additional $67.5 million upon achievement of specified development and regulatory milestones, and up to $165.5 million upon achievement of certain commercial milestones. Forma is also entitled to receive tiered royalties on net sales of licensed products at percentages ranging from the low-teens to mid-thirties, as well as certain portion of sublicensing revenue, subject to certain standard reductions and offsets.
The transaction was accounted for as an acquisition of asset under ASC 730, Research and Development. Milestone payments incurred prior to regulatory approval of an indication associated with the acquired licensed asset were recognized as research and development expense. Accordingly, the upfront fee of $2.0 million was recorded as acquired in-process research and development (IPR&D) and expensed within research and development expense in the statements of operations in 2022. In addition, a $2.5 million regulatory milestone achieved prior to the FDA approval of REZLIDHIA in December 2022 was also recognized as research and development expense in 2022.
On December 1, 2022, the FDA approved REZLIDHIA capsules for the treatment of adult patients with R/R AML with susceptible IDH1 mutations, as detected by an FDA-approved test. Following approval and first commercial sale in December 2022, Forma became entitled to receive aggregate milestone payments of $15.0 million. Because these milestone obligations were incurred upon and after regulatory approval, the amounts were capitalized as intangible assets on our condensed balance sheet. The intangible assets are amortized on a straight-line basis over an estimated useful life of 14 years, with amortization recognized in cost of product sales. Royalties are also recognized in cost of product sales, as the related product sales occur.
6. Stock-Based Compensation
Stock-based compensation for the periods presented was as follows (in thousands):
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| Selling, general and administrative | $ | 3,015 | | | $ | 2,452 | | | | | |
| Research and development | 441 | | | 872 | | | | | |
| Total stock-based compensation expense | $ | 3,456 | | | $ | 3,324 | | | | | |
Pursuant to our 2018 Equity Incentive Plan (2018 Plan) and our Inducement Plan, as amended (Inducement Plan, and together with 2018 Plan, the Equity Incentive Plans), during the three months ended March 31, 2026, we granted stock options to purchase 36,775 shares of common stock, with weighted-average grant-date fair value of $30.14 per share, and 618,358 RSUs, with a grant-date weighted-average fair value of $35.77 per share. In recent years, we have increased our use of RSUs, and beginning in 2026, RSUs represent the majority of our equity awards. Beginning in 2026, we also grant performance-based RSUs tied to the achievement of specified corporate performance milestones. These equity awards generally vest over a three-year period, except for performance-based awards, which vest upon the achievement of specified corporate performance milestones.
The fair value of the RSU is based on the market price of our common stock on the date of grant. The fair value of stock option awards is estimated on the grant date using the Black-Scholes option pricing model. The following table summarizes the weighted-average assumptions relating to stock options granted during the periods presented:
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| Risk-free interest rate | 3.8 | % | | 4.4 | % | | | | |
| Expected term (in years) | 5.3 | | 6.5 | | | | |
| Dividend yield | 0.0 | % | | 0.0 | % | | | | |
| Expected volatility | 90.5 | % | | 88.5 | % | | | | |
During the three months ended March 31, 2026, 121,846 stock options were exercised and 226,221 RSUs were released. During the three months ended March 31, 2026, we withheld 105,728 shares of common stock, with an aggregate value of approximately $3.7 million, in connection with the net share settlement of RSUs to satisfy employees’ minimum statutory tax withholding obligations upon vesting. The withheld shares were valued based on the closing market price of our common stock on the applicable vesting dates. Such share withholdings are treated as share repurchases for accounting purposes and are reflected as a reduction to additional paid-in capital.
As of March 31, 2026, there were 3,436,088 stock options and 983,847 RSUs outstanding. Of these, 168,564 stock options and 67,200 RSUs were performance-based awards for which achievement of the related corporate milestones
was deemed not probable as of March 31, 2026. Accordingly, none of the associated $5.5 million of grant date fair value has been recognized as stock-based compensation expense as of March 31, 2026.
As of March 31, 2026, there was approximately $33.4 million of unrecognized stock-based compensation expense, which is expected to be recognized over a remaining weighted-average period of 2.52 years. This amount relates to time-based stock options and RSUs, as well as performance-based stock options and RSUs for which achievement of the related corporate performance milestones was considered probable.
As of March 31, 2026, there were 702,279 shares of common stock available for future grant under our Equity Incentive Plans.
Employee Stock Purchase Plan
The current 24-month offering under our Employee Stock Purchase Plan (Purchase Plan) commenced on July 1, 2024. As of March 31, 2026, there was approximately $0.2 million of unrecognized stock-based compensation cost related to the Purchase Plan, which is expected to be recognized over a remaining weighted-average period of 0.25 years. As of March 31, 2026, 96,998 shares were available for future issuance under the Purchase Plan.
7. Other Balance Sheet Components
Inventories
Inventories for the periods presented consist of the following (in thousands):
| | | | | | | | | | | |
| As of |
| March 31, 2026 | | December 31, 2025 |
| Raw materials | $ | 3,427 | | | $ | 4,514 | |
| Work in process | 6,229 | | | 3,910 | |
| Finished goods | 4,097 | | | 4,712 | |
| Total | $ | 13,753 | | | $ | 13,136 | |
| Reported as: | | | |
| Inventories | $ | 11,652 | | | $ | 11,506 | |
| Other assets | 2,101 | | | 1,630 | |
| Total | $ | 13,753 | | | $ | 13,136 | |
Non-current inventories included within other assets in the condensed balance sheet consist primarily of active pharmaceutical ingredient (API) classified as raw materials which have multi-year shelf life, as well as certain work in process and finished goods inventories that are not expected to be consumed beyond our normal operating cycle.
Advance payments to our contract manufacturers to manufacture APIs as well as APIs pending final release for commercial usage are classified as prepaid inventory and included within prepaid and other current assets in the condensed balance sheet. See prepaid and other current assets below for related details.
Prepaid and other current assets
Prepaid and other current assets for the periods presented consist of the following (in thousands):
| | | | | | | | | | | |
| As of |
| March 31, 2026 | | December 31, 2025 |
| Prepaid inventory | $ | 13,407 | | | $ | 11,849 | |
| Prepaid research and development costs | 5,204 | | | 4,886 | |
| Others | 7,135 | | | 5,207 | |
| Total prepaid and other current assets | $ | 25,746 | | | $ | 21,942 | |
Intangible assets
Intangible assets consist of the following (in thousands):
| | | | | | | | | | | |
| As of |
| March 31, 2026 | | December 31, 2025 |
| Intangible assets cost | $ | 30,360 | | | $ | 30,360 | |
| Accumulated amortization | (6,200) | | | (5,612) | |
| Intangible assets, net | $ | 24,160 | | | $ | 24,748 | |
Amortization expense included in cost of product sales in the condensed statements of operations was $0.6 million for each of the three months ended March 31, 2026 and 2025.
The following table presents the estimated future amortization expense of intangible assets as of March 31, 2026 (in thousands):
| | | | | |
| Remainder of 2026 | $ | 1,763 | |
| 2027 | 2,351 | |
| 2028 | 2,351 | |
| 2029 | 2,351 | |
| 2030 | 2,351 | |
| Thereafter | 12,993 | |
| $ | 24,160 | |
8. Cash, Cash Equivalents, Restricted Cash, and Short-Term Investments
Cash, cash equivalents, restricted cash, and short-term investments for the periods presented consist of the following (in thousands):
| | | | | | | | | | | |
| As of |
| March 31, 2026 | | December 31, 2025 |
| Cash | $ | 10,719 | | | $ | 20,020 | |
| Restricted cash | 57 | | | 57 | |
| Money market funds | 12,593 | | | 14,509 | |
| US treasury bills | 76,474 | | | 63,994 | |
| Government-sponsored enterprise securities | 23,113 | | | 20,692 | |
| Corporate bonds and commercial paper | 23,785 | | | 35,740 | |
| $ | 146,741 | | | $ | 155,012 | |
| Reported as: | | | |
| Cash and cash equivalents | $ | 24,411 | | | $ | 40,580 | |
| Short-term investments | 122,273 | | | 114,375 | |
| Restricted cash reported within other assets | 57 | | | 57 | |
| $ | 146,741 | | | $ | 155,012 | |
Cash equivalents and short-term investments include the following securities with gross unrealized gains and losses (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| As of March 31, 2026 | | Amortized
Cost | | Gross
Unrealized
Gains | | Gross
Unrealized
Losses | | Fair Value |
| US treasury bills | | $ | 76,554 | | | $ | 30 | | | $ | (110) | | | $ | 76,474 | |
| Government-sponsored enterprise securities | | 23,147 | | | 8 | | | (42) | | | 23,113 | |
| Corporate bonds and commercial paper | | 23,799 | | | 4 | | | (18) | | | 23,785 | |
| Total | | $ | 123,500 | | | $ | 42 | | | $ | (170) | | | $ | 123,372 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| As of December 31, 2025 | | Amortized
Cost | | Gross
Unrealized
Gains | | Gross
Unrealized
Losses | | Fair Value |
| US treasury bills | | $ | 63,868 | | | $ | 126 | | | $ | — | | | $ | 63,994 | |
| Government-sponsored enterprise securities | | 20,663 | | | 30 | | | (1) | | | 20,692 | |
| Corporate bonds and commercial paper | | 35,714 | | | 28 | | | (2) | | | 35,740 | |
| Total | | $ | 120,245 | | | $ | 184 | | | $ | (3) | | | $ | 120,426 | |
Our short-term investments are classified as available-for-sale securities. Accordingly, we have classified these securities as short-term investments on our condensed balance sheets as they are available for use in the current operations. Accrued interest receivable, included within prepaid and other assets, was $1.0 million and $0.9 million of March 31, 2026 and December 31, 2025, respectively. As of March 31, 2026 and December 31, 2025, our cash equivalents and short-term investments had a weighted-average time to maturity of approximately 317 days and 303 days, respectively.
As of March 31, 2026, a total of 67 individual securities had been in an unrealized loss position for 12 months or less, and the losses were determined to be temporary. We regularly review the securities in an unrealized loss position and evaluate the current expected credit loss by considering factors such as historical experience, market data, issuer-specific factors, and current economic conditions. We have not recognized any credit losses as of March 31, 2026 and December 31, 2025.
The following table shows the fair value and gross unrealized losses of our investments in individual securities that are in an unrealized loss position, aggregated by investment category (in thousands):
| | | | | | | | | | | | | | |
| As of March 31, 2026 | | Fair Value | | Gross Unrealized Losses |
| US treasury bills | | $ | 11,023 | | | $ | (37) | |
| Government-sponsored enterprise securities | | 11,645 | | | (40) | |
| Corporate bonds and commercial paper | | 72,607 | | | (93) | |
| Total | | $ | 95,275 | | | $ | (170) | |
9. Fair Value
The table below summarizes the fair value of our cash equivalents and short-term investments measured at fair value on a recurring basis, and are categorized based upon the lowest level of significant input to the valuations (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Assets at Fair Value as of March 31, 2026 |
| Level 1 | | Level 2 | | Level 3 | | Total |
| Money market funds | $ | 12,593 | | | $ | — | | | $ | — | | | $ | 12,593 | |
| US treasury bills | — | | | 76,474 | | | — | | | 76,474 | |
| Government-sponsored enterprise securities | — | | | 23,113 | | | — | | | 23,113 | |
| Corporate bonds and commercial paper | — | | | 23,785 | | | — | | | 23,785 | |
| Total | $ | 12,593 | | | $ | 123,372 | | | $ | — | | | $ | 135,965 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| Assets at Fair Value as of December 31, 2025 |
| Level 1 | | Level 2 | | Level 3 | | Total |
| Money market funds | $ | 14,509 | | | $ | — | | | $ | — | | | $ | 14,509 | |
| US treasury bills | — | | | 63,994 | | | — | | | 63,994 | |
| Government-sponsored enterprise securities | — | | | 20,692 | | | — | | | 20,692 | |
| Corporate bonds and commercial paper | — | | | 35,740 | | | — | | | 35,740 | |
| Total | $ | 14,509 | | | $ | 120,426 | | | $ | — | | | $ | 134,935 | |
10. Debt
The following table summarizes loans payable, net (in thousands):
| | | | | | | | | | | |
| As of |
| March 31, 2026 | | December 31, 2025 |
| Principal outstanding | $ | 45,000 | | | $ | 52,500 | |
| Unamortized debt issuance costs | (144) | | | (206) | |
| Principal outstanding, net of unamortized debt issuance costs | $ | 44,856 | | | $ | 52,294 | |
| Reported as: | | | |
| Loans payable, net, current portion | $ | 29,865 | | | $ | 29,812 | |
| Long-term portion of loans payable, net | 14,991 | | | 22,482 | |
| $ | 44,856 | | | $ | 52,294 | |
The outstanding loans payable as of the periods presented were related to our Credit and Security Agreement (Credit Agreement) with MidCap Financial Trust (MidCap) entered into on September 27, 2019 (Closing Date) and amended on March 29, 2021 (First Amendment), February 11, 2022 (Second Amendment), July 27, 2022 (Third
Amendment), and on April 11, 2024 (Fourth Amendment). The Credit Agreement provided for a $60.0 million term loan credit facility.
Under the Credit Agreement, as amended, the term loans were scheduled to mature on September 1, 2027, and the interest-only period was through October 1, 2025. The term loans bore interest equal to the sum of one-month Secured Overnight Financing Rate (SOFR) plus an adjustment of 0.11448%, subject to a 4.00% applicable floor, plus applicable margin of 6.50%. A final payment fee of 4.25% of principal was due at maturity date.
We could make voluntary prepayments, in whole or in part, subject to certain prepayment premiums and additional interest payments. The Credit Agreement also contained certain provisions, such as event of default and change in control provisions, which, if triggered, would have required us to make mandatory prepayments on the term loan, which are subject to certain prepayment premiums and additional interest payments. The obligations under the amended Credit Agreement were secured by a perfected security interest in all of our assets including our intellectual property.
Interest expense, including amortization of the debt discount and accretion of the final fees related to the Credit Agreement for the three months ended March 31, 2026 and 2025 was $1.4 million and $1.9 million, respectively. Accrued interest of $2.6 million and $2.6 million was included within other accrued liabilities in the condensed balance sheet as of March 31, 2026 and December 31, 2025, respectively.
The following table presents the future minimum principal payments of the outstanding loan as of March 31, 2026 (in thousands):
| | | | | |
| Remainder of 2026 | $ | 22,500 | |
| 2027 | 22,500 | |
| Principal amount (Tranches 1, 2, 3 and 4) | $ | 45,000 | |
The amended Credit Agreement contained certain covenants which, among others, required us to deliver financial reports at designated times of the year and maintain minimum unrestricted cash and trailing net revenues. As of March 31, 2026, we were not in violation of any covenants.
On May 5, 2026, we terminated the Credit Agreement and repaid all outstanding term loans thereunder. Concurrently, we entered into a new Credit and Security Agreement with MidCap providing for a revolving credit facility (new Credit Agreement). See “Note 14 - Subsequent Events” for additional information.
11. Commitments and Contingencies
Operating Leases
We lease our headquarters facility in South San Francisco, California under a lease agreement that expires in July 2027. Operating lease expense was $0.2 million for each of the three months ended March 31, 2026 and 2025. Cash paid for amounts included in the measurement of operating lease liabilities was $0.2 million for each of the three months ended March 31, 2026 and 2025.
As of March 31, 2026, the weighted average remaining term was 1.33 years, and future minimum lease payments were approximately $0.9 million.
Purchase Commitments and Obligations
In the ordinary course of business, we enter into agreements with contract manufacturers to manufacture our inventory products. These agreements generally include termination provisions that may require us to pay cancellation fees, which vary depending on the timing of termination and may equal up to the full value of the work order. In October 2024, we entered into an agreement with a third-party contract manufacturer to manufacture TAVALISSE, with deliveries expected from 2026 through 2029. As of March 31, 2026, the contractual obligation not included in our financial statements related to an agreement that may potentially be subjected to cancellation fees were approximately $19.2 million. Of this amount, approximately $5.4 million is expected to be due in the remainder of 2026, and $9.5 million is expected to be due in 2027 and 2028. As of March 31, 2026, we have not incurred any cancellation fees under our agreements with contract manufacturers.
Legal Contingencies
From time to time, we may become involved in legal proceedings arising in the ordinary course of our business. We are not presently a party to any material legal proceedings that, if determined adversely to us, would have a material adverse effect on our business, financial condition, results of operations, or cash flows.
12. Income Taxes
The following table presents provision for income tax for the periods presented (in thousands):
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| Provision for income taxes | $ | 3,003 | | | $ | 65 | | | | | |
The quarterly provision for income taxes is determined by applying the estimated annual effective tax rate to the year-to-date pre-tax income, adjusted for any discrete items. The estimated annual effective tax rate is updated at the end of each reporting period.
The provision for income taxes for the three months ended March 31, 2026 primarily consisted of federal income tax expense of $2.4 million and estimated state income taxes of $0.6 million. Prior to the fourth quarter of 2025, we maintained a full valuation allowance against our deferred tax assets. Although we do not expect to incur federal cash income taxes due to sufficient net operating loss (NOL) and research and development credit carryforwards, we recognized federal income tax expense based on the estimated impact of utilizing the deferred tax assets associated with such carryforwards. The total tax expense differs from the amount computed at the federal statutory rate primarily due to certain non-deductible expenses and state income taxes.
For the three months ended March 31, 2025, the provision for income taxes primarily consisted of estimated state income taxes. The tax expense differs from the amount computed at the federal statutory rate primarily due to the impact of the valuation allowance and state taxes.
13. Segment Information
We view our operations and manage our business as one operating segment, and our chief operating decision maker (CODM) is our chief executive officer. The following table presents segment information for the periods presented (in thousands):
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| | | |
| Total Revenues | $ | 58,818 | | | $ | 53,333 | | | | | |
| Less: | | | | | | | |
| Employee related expenses | 21,002 | | | 19,538 | | | | | |
| Commercial related expenses | 7,040 | | | 5,339 | | | | | |
| Outside clinical trial related expenses | 6,927 | | | 4,371 | | | | | |
| Cost of product sales | 4,606 | | | 4,409 | | | | | |
| Consultants and third-party services | 4,006 | | | 3,409 | | | | | |
| Other segment items | 3,352 | | | 3,494 | | | | | |
| Interest expense, net | 228 | | | 1,262 | | | | | |
| Provision for income taxes | 3,003 | | | 65 | | | | | |
| Segment income | $ | 8,654 | | | $ | 11,446 | | | | | |
There are no reconciling items or adjustments between segment income presented above and net income as presented in our statements of operations. The CODM does not review assets in evaluating the segment performance, accordingly, such information is not presented.
For details of revenues disaggregated by category, see “Note 3 – Revenues.”
Employee related expenses primarily consist of salaries, employee benefits, other employee related expenses and stock-based compensation expense. For additional details of stock-based compensation expense, see “Note 6 – Stock-Based Compensation.” Other segment items for the periods presented primarily consist of travel related expenses, business insurance, taxes and licenses, and subscription services.
14. Subsequent Events
Credit Agreement with MidCap
On May 5, 2026, we terminated our Credit Agreement with MidCap, which provided for a $60.0 million term loan credit facility. In connection with the termination, we repaid all outstanding borrowings under the term loan facility, including applicable prepayment premiums, accrued interest and final payment fees.
Concurrently, we entered into a new Credit Agreement with MidCap providing for a revolving credit facility with a maximum borrowing capacity of $40.0 million, with an option to increase the facility to $60.0 million, subject to customary conditions. Availability under the revolving credit facility is subject to a borrowing base based primarily on eligible accounts receivable and inventory. The revolving credit facility has a five-year term and bears interest at a rate equal to one-month SOFR, subject to a 2.00% floor, plus an applicable margin of 4.00%.
The obligations under the revolving credit facility are secured by a first-priority security interest in substantially all of our assets, including our intellectual property. The revolving credit facility includes customary fees, including an unused commitment fee, administrative fee and prepayment premiums during the initial period. As of the date of this filing, we had an outstanding borrowing of $8.0 million under the revolving credit facility.
The revolving credit facility contains customary covenants that, among other things, require us to deliver financial reports at specified times and maintain minimum liquidity and trailing twelve month product revenue. The minimum product revenue covenant is tested only during periods when the liquidity falls below specified thresholds.
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations
This discussion and analysis should be read in conjunction with our financial statements and the accompanying notes included in this report and the audited financial statements and accompanying notes included in our Annual Report on Form 10-K for the year ended December 31, 2025 filed with the SEC on March 3, 2026. Our financial results for the three months ended March 31, 2026 are not necessarily indicative of results that may occur in future interim periods or for the full fiscal year.
This Quarterly Report on Form 10-Q contains statements indicating expectations about future performance and other forward-looking statements within the meaning of Section 27A of the Securities Act and Section 21E of the Securities Exchange Act of 1934, as amended (Exchange Act), and the Private Securities Litigation Reform Act of 1995, that involve risks and uncertainties. We usually use words such as “may,” “will,” “would,” “should,” “could,” “expect,” “plan,” “anticipate,” “might,” “believe,” “estimate,” “predict,” “intend,” or the negative of these terms or similar expressions to identify these forward-looking statements. These statements appear throughout this Quarterly Report on Form 10-Q and are statements regarding our current expectations, beliefs or intent, primarily with respect to our operations and related industry developments. Examples of these statements include, but are not limited to: our business and scientific strategies; risks and uncertainties associated with the commercialization, distribution, marketing, and payment for our products in the US and outside the US; risks that the FDA, EMA, the Medicines and Health Products Regulatory Agency (MHRA) or other regulatory authorities may make adverse decisions regarding our products; the impact of the US federal government shutdowns or agency funding disruptions; the progress of our and our collaborators’ product development programs, including clinical testing, and the timing of results thereof; our corporate collaborations and revenues that may be received from our collaborations and the timing of those potential payments; our expectations with respect to obligations to entities party to commercial or licensing agreements with us and the timing of those obligations; our expectations with respect to timing of recognizing product sales; our expectations with respect to the volume of product sales; our expectations with respect to regulatory submissions and approvals; our drug discovery technologies; our research and development expense; protection of our intellectual property and our intention to vigorously enforce our intellectual property rights; the availability and sufficiency of our cash and capital resources and the need for additional capital; our ability to successfully identify and acquire or in-license products or companies; our operations and legal risks; and the effectiveness of our cybersecurity risk management process. You should not place undue reliance on these forward-looking statements. Our actual results could differ materially from those anticipated in these forward-looking statements for many reasons, including as a result of the risks and uncertainties discussed under the heading “Risk Factors” in Item 1A of Part II of this Quarterly Report on Form 10-Q. Any forward-looking statement speaks only as of the date on which it is made, and we undertake no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events, except as required by applicable law. New factors emerge from time to time, and it is not possible for us to predict which factors will arise. In addition, we cannot assess the impact of each factor on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements.
Overview
We are a biotechnology company dedicated to developing and providing novel therapies that significantly improve the lives of patients with hematologic disorders and cancer. We focus on products that address signaling pathways that are critical to disease mechanisms.
TAVALISSE (fostamatinib disodium hexahydrate) is our first FDA-approved product and is the only approved oral SYK inhibitor for the treatment of adult patients with chronic ITP who have had an insufficient response to a previous treatment. The product is also commercially available in Europe and the UK (as TAVLESSE), and in Japan, Korea, Canada and Israel (as TAVALISSE) for the treatment of chronic ITP in adult patients.
REZLIDHIA (olutasidenib) is our second FDA-approved product indicated for the treatment of adult patients with R/R AML with a susceptible IDH1 mutation as detected by an FDA-approved test. We in-licensed REZLIDHIA from Forma with exclusive, worldwide rights for its development, manufacturing and commercialization, pursuant to a license and transition services agreement entered in July 2022.
GAVRETO (pralsetinib) is our third FDA-approved product which we began commercializing in June 2024. GAVRETO is a once daily, small molecule, oral, kinase inhibitor of wild-type RET and oncogenic RET fusions. GAVRETO is approved by the FDA for the treatment of adult patients with metastatic RET fusion-positive NSCLC as detected by an FDA-approved test. GAVRETO is also approved under accelerated approval based on overall response rate and duration response rate, for the treatment of adult and pediatric patients 12 years of age and older with advanced or metastatic RET fusion-positive thyroid cancer who require systemic therapy and who are radioactive iodine-refractory (if radioactive iodine is appropriate). We acquired the rights to research, develop, manufacture and commercialize GAVRETO in the US from Blueprint pursuant to an asset purchase agreement entered in February 2024.
Our development pipeline includes R289, our dual IRAK1/4 inhibitor program, which is being advanced in an open-label, Phase 1b study to determine the safety, tolerability and preliminary efficacy of the drug in patients with lower-risk MDS who are relapsed, refractory or resistant to prior therapies.
To expand our evaluation of olutasidenib in other disease areas with IDH1 mutations, we have strategic development collaborations with MDACC and with CONNECT.
Business Updates
Commercial Products
TAVALISSE net product sales for the three months ended March 31, 2026 were $37.3 million, an increase of $8.8 million, or 31%, compared to $28.5 million for the same period in 2025. The increase was primarily driven by higher volumes and higher price per bottle, as well as a favorable impact from lower revenue reserves.
REZLIDHIA net product sales for the three months ended March 31, 2026 were $8.0 million, an increase of $1.9 million, or 31%, compared to $6.1 million for the same period in 2025. The increase was primarily driven by higher volumes and higher price per bottle, partially offset by higher revenue reserves.
GAVRETO net product sales for the three months ended March 31, 2026 were $9.6 million, an increase of $0.6 million, or 7%, compared to $9.0 million for the same period in 2025. The increase was primarily driven by higher price per bottle and, to a lesser extent, higher volumes.
R289, an Oral IRAK1/4 Inhibitor for Lower-Risk MDS
We advanced the development of our dual IRAK1/4 inhibitor program, following evaluation of single and multiple ascending doses of R289 in healthy subjects. The ongoing Phase 1b open-label, multicenter study evaluates the safety, tolerability and preliminary efficacy of R289 in patients with R/R lower-risk MDS. This Phase 1b study is expected to enroll approximately 86 patients (up to 36 patients in the dose escalation phase, up to 40 patients in the dose expansion phase, and 10 patients in an exploratory cohort evaluating post- or ineligible erythropoiesis-stimulating agent (ESA), treatment naïve patients). The primary objective of the study is safety, with secondary and exploratory objectives to assess preliminary efficacy and characterize the pharmacokinetic and pharmacodynamic profile of R289. Enrollment in the dose escalation part of the study was completed in July 2025. In October 2025, we announced enrollment of the first patient in the dose expansion part of the study, where up to 40 patients will be randomized to receive either 500 mg once daily or twice daily to determine the recommended Phase 2 dose for future clinical studies. Enrollment is ongoing and we expect to
complete enrollment of the dose expansion phase of the Phase 1b study and select the recommended Phase 2 dose for future clinical studies in the second half of 2026. We anticipate sharing preliminary data from the dose expansion phase of the study by the end of 2026.
Olutasidenib in AML, Other Hematologic Cancers and HGG
We have a Strategic Collaboration Agreement with MDACC, a comprehensive cancer research, treatment, and prevention center. The collaboration expanded our evaluation of olutasidenib in AML and other hematologic cancers with IDH1 mutations. Under the Strategic Collaboration Agreement, we jointly lead the clinical development efforts with MDACC to evaluate the potential of olutasidenib to treat newly diagnosed and R/R patients with AML, higher-risk MDS, and advanced myeloproliferative neoplasms, in combination with other agents. The collaboration also supports the evaluation of olutasidenib as monotherapy in patients with IDH1 mutated clonal cytopenia of undetermined significance (CCUS) and lower-risk MDS, as well as maintenance therapy following hematopoietic stem cell transplant. Further, this collaboration also supports the evaluation of olutasidenib in combination with co-targeted therapies in patients with R/R IDH1-mutated myeloid malignancies harboring activated signaling pathway mutations. There are five studies open for enrollment associated with the multi-year strategic development alliance. Under the Strategic Collaboration Agreement, we will provide MDACC the study materials and $15.0 million in time-based milestone payments as compensation for services to be provided for the studies, over the five-year collaboration term, unless terminated earlier as provided for in the agreement. Through March 31, 2026, we provided $5.3 million funding to MDACC.
We also have a collaboration with CONNECT, an international collaborative network of pediatric cancer centers, to conduct a Phase 2 clinical trial to evaluate olutasidenib in combination with temozolomide in patients with HGG harboring an IDH1 mutation. Under the collaboration, CONNECT will include the olutasidenib treatment arm within CONNECT’s TarGet study, a molecularly guided Phase 2 umbrella clinical trial for HGG. In our sponsored arm, TarGet-D, adolescents and young adult patients (ages 12 to 39 years old) with newly-diagnosed IDH1-mutation positive HGG will receive maintenance therapy with olutasidenib in combination with temozolomide for the first year after radiotherapy, followed by olutasidenib monotherapy for the second year. Under the collaboration, we will provide CONNECT with funding up to $3.0 million and study material over the four-year collaboration. Enrollment in the Phase 2 TarGet-D study is ongoing.
Global Strategic Partnership with Lilly
We entered into a global exclusive and strategic collaboration with Lilly in February 2021 to develop and commercialize ocadusertib (previously R552), an investigational, potent and selective RIPK1 inhibitor, for the treatment of non-CNS diseases, and additional RIPK1 inhibitors for the treatment of CNS diseases. RIPK1 is implicated in a broad range of key inflammatory cellular processes and plays a key role in tumor necrosis factor signaling, especially in the induction of pro-inflammatory necroptosis.
On April 16, 2026, we received written notice from Lilly of its decision to terminate the Lilly Agreement, which will become effective June 15, 2026. Following termination of the Lilly Agreement, including the prior termination of the CNS disease program effective in November 2025, we do not expect to receive future milestones or royalties under the Lilly Agreement. Pursuant to such termination, the Lilly Agreement will terminate in accordance with its terms, including the cessation of Lilly’s rights to the licensed compounds, subject to any applicable transition provisions. We expect to regain full rights to the licensed compounds and related programs upon termination.
Credit Agreement with MidCap
On May 5, 2026, we terminated our Credit Agreement with MidCap, which provided for a $60.0 million term loan facility, under which $45.0 million was outstanding as of March 31, 2026, and repaid all outstanding borrowings thereunder, including applicable fees and expenses. Concurrently, we entered into a new Credit Agreement with MidCap, which provides for a revolving credit facility with an initial borrowing capacity of $40.0 million and an option to increase to $60.0 million, subject to customary conditions. Availability under the revolving credit facility is subject to a borrowing base based primarily on eligible accounts receivable and inventory. The revolving credit facility under the new Credit Agreement has a five-year term and bears interest at a rate equal to one-month SOFR, subject to a 2.00% floor, plus an applicable margin of 4.00%. The obligations under the revolving credit facility are secured by a first-priority security interest in substantially all of our assets, including our intellectual property. The revolving credit facility includes customary fees, including an unused commitment fee, administrative fee and prepayment premiums during the initial period. As of the date of this filing, we had an outstanding borrowing of $8.0 million under the revolving credit facility.
Our Product Portfolio
The following table summarizes our portfolio:
Commercial Products
TAVALISSE/Fostamatinib in ITP
TAVALISSE overview
Chronic ITP affects an estimated 81,300 adult patients in the US. In patients with ITP, the immune system attacks and destroys the body’s own blood platelets, which play an active role in blood clotting and healing. ITP patients can suffer extraordinary bruising, bleeding and fatigue as a result of low platelet counts. Current therapies for ITP include steroids, blood platelet production boosters that imitate thrombopoietin (TPO) and splenectomy.
Taken in tablet form, fostamatinib blocks the activation of SYK inside immune cells. ITP is typically characterized by the body producing antibodies that attach to healthy platelets in the blood stream. Immune cells recognize these antibodies and affix to them, which activates the SYK enzyme inside the immune cell, and triggers the destruction of the antibody and the attached platelet. When SYK is inhibited by fostamatinib, it interrupts this immune cell function and allows the platelets to escape destruction. The results of our Phase 2 clinical trial, in which fostamatinib was orally administered to 16 adults with chronic ITP, published in Blood, showed that fostamatinib significantly increased the platelet counts of certain ITP patients, including those who had failed other currently available agents.
Our Fostamatinib for Immune Thrombocytopenia (FIT) Phase 3 clinical program had a total of 150 ITP patients who were randomized into two identical multicenter, double-blind, placebo-controlled clinical trials. The patients were diagnosed with persistent or chronic ITP, and had blood platelet counts consistently below 30,000 per microliter of blood. Two-thirds of the subjects received fostamatinib orally at 100 mg twice daily (bid) and the other third received placebo on the same schedule. Subjects were expected to remain on treatment for up to 24 weeks. At week four of treatment, subjects who failed to meet certain platelet counts and met certain tolerability thresholds could have their dosage of fostamatinib (or corresponding placebo) increased to 150 mg bid. The primary efficacy endpoint of this program was a stable platelet response by week 24 with platelet counts at or above 50,000 per microliter of blood for at least four of the final six qualifying blood draws. In August 2016, we announced the results of the first FIT study, reporting that fostamatinib met the study’s primary efficacy endpoint. The study showed that 18% of patients receiving fostamatinib achieved a stable platelet response compared to none receiving a placebo control. In October 2016, we announced the results of the second FIT study, reporting that the response rate (16% in the treatment group, versus 4% in the placebo group) was consistent with the first study, although the difference was not statistically significant. In the ITP double-blind studies, the most
commonly reported adverse reactions occurring in at least 5% of patients treated with TAVALISSE were diarrhea, hypertension, nausea, dizziness, increased alanine aminotransferase, increased aspartate aminotransferase, respiratory infection, rash, abdominal pain, fatigue, chest pain, and neutropenia. Serious adverse drug reactions occurring in at least 1% of patients treated with TAVALISSE in the ITP double-blind studies were febrile neutropenia, diarrhea, pneumonia, and hypertensive crisis. A post-hoc analysis from our Phase 3 clinical program in adult patients with chronic ITP, highlighting the potential benefit of using TAVALISSE in earlier lines of therapy, was published in the British Journal of Haematology in July 2020. In addition, a report describing the long-term safety and durable efficacy of TAVALISSE with up to five years of treatment was published in Therapeutic Advances in Hematology in 2021.
The FDA granted orphan drug designation for fostamatinib for the treatment of ITP in August 2015. TAVALISSE was approved by the FDA in April 2018 for the treatment of ITP in adult patients who have had an insufficient response to a previous treatment, and successfully launched in the US in May 2018.
Competitive landscape for TAVALISSE
Our industry is intensely competitive and subject to rapid and significant technological change. TAVALISSE is competing with other existing therapies. In addition, a number of companies are pursuing the development of pharmaceuticals that target the same diseases and conditions that we are targeting. For example, there are existing therapies and drug candidates in development for the treatment of ITP that may be alternative therapies to TAVALISSE.
Currently, corticosteroids remain the most common first line therapy for ITP, occasionally in conjunction with intravenous immunoglobulin (IVIg) or anti-Rh(D) to help further augment platelet count recovery, particularly in emergency situations. However, it has been estimated that frontline agents lead to durable remissions in only a small percentage of newly diagnosed adults with ITP. Moreover, concerns with steroid-related side effects often restrict therapy to approximately four weeks. As such, many patients progress to persistent or chronic ITP, requiring other forms of therapeutic intervention. In long-term treatment of chronic ITP, patients are often cycled through several therapies over time in order to maintain a sufficient response to the disease.
Other approaches to treat ITP are varied in their mechanism of action, and there is no consensus about the sequence of their use. Options include splenectomy, thrombopoietin receptor agonists (TPO-Ras) and various immunosuppressants (such as rituximab). The response rate criteria of the above-mentioned options vary, precluding a comparison of response rates for individual therapies.
Even with the above treatment options, a significant number of patients remain severely thrombocytopenic for long durations and are subject to risk of spontaneous or trauma-induced hemorrhage. The addition of fostamatinib to the currently available treatment options could be beneficial because it has a different mechanism of action than any of the therapies that are currently available. Fostamatinib is a potent and relatively selective SYK inhibitor, and its inhibition of Fc receptors and B-cell receptors of signaling pathways make it a potentially broad immunomodulatory agent.
The FDA recently approved the product WAYRILZTM (Sanofi SA) for the treatment of adults with persistent or chronic ITP. Other products in the US that are approved by the FDA to increase platelet production through binding to TPO receptors on megakaryocyte precursors include PROMACTA® (Novartis International AG), Nplate® (Amgen, Inc.), DOPTELET® (Swedish Orphan Biovitrum AB) and ALVAIZTM (Teva Pharmaceutical Industries Ltd.). In addition, the availability of generic versions of TPO receptor agonists may further intensify competition and adversely affect the market. In the longer term, we may eventually face competition from potential manufacturers of generic versions of our marketed products, including the proposed generic version of TAVALISSE, which, if approved and allowed to enter the market, could result in significant decreases in the revenue derived from the sale of TAVALISSE and thereby materially harm our business and financial condition.
TAVALISSE Commercial activities, including sales and marketing
Our marketing and sales efforts are focused on hematologists and hematologist-oncologists in the US who manage chronic adult ITP patients. We have a fully integrated commercial team consisting of sales, marketing, market access, and commercial operations functions. Our sales team promotes our products in the US using customary pharmaceutical company practices. Our products are sold initially through third-party wholesale distribution and specialty pharmacy channels and group purchasing organizations before being ultimately prescribed to patients. To facilitate our commercial activities in the US, we also enter into arrangements with various third parties, including advertising agencies, market research firms and other sales-support-related services as needed. We believe that our commercial team and distribution
practices are adequate to ensure that our marketing efforts reach relevant customers and deliver our products to patients in a timely and compliant fashion. Also, to help ensure that all eligible patients in the US have appropriate access to our products, we have established a reimbursement and patient support program called Rigel OneCare ® (ROC). Through ROC, we provide co-pay assistance to qualified, commercially insured patients to help minimize out-of-pocket costs and provide free products to uninsured or under-insured patients who meet certain established clinical and financial eligibility criteria. In addition, ROC is designed to provide reimbursement support, such as information related to prior authorizations, benefits investigations and appeals.
We have entered into various license and commercial agreements to commercialize fostamatinib globally as discussed below, but we retain the global rights to fostamatinib outside of the respective territories under such license and commercial agreements.
Fostamatinib outside of the US
We have a commercialization license agreement with Grifols for exclusive rights to commercialize fostamatinib for human diseases, and non-exclusive rights to develop fostamatinib in their territory. Grifols territory includes European Union (EU), the UK, Turkey, the Middle East, North Africa and Russia (including CIS). In January 2020, the European Commission (EC) granted a centralized MA for fostamatinib (TAVLESSE) valid throughout the EU and which has been grandfathered in the UK, after the departure of the UK from the EU, for the treatment of chronic ITP in adult patients who are refractory to other treatments. Grifols has launched TAVLESSE in the UK and certain countries in EU including Germany, France, Italy and Spain, and continues a phased rollout across the rest of EU.
We have an exclusive license and supply agreement with Kissei to develop and commercialize fostamatinib in all current and potential indications in Japan, China, Taiwan and Korea. Kissei is a Japan-based pharmaceutical company addressing patients’ unmet medical needs through its research, development and commercialization efforts, as well as through collaborations with partners. Japan has the third highest prevalence of chronic ITP in the world behind the US and Europe. Kissei was granted orphan drug designation from the Japanese Ministry of Health, Labor and Welfare for R788 (fostamatinib) in chronic ITP in February 2020. In December 2022, Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) approved TAVALISSE for the treatment of persistent and chronic ITP, and in April 2023, Kissei launched TAVALISSE for chronic ITP in Japan. In January 2025, Kissei announced the Korean Ministry of Food and Drug Safety approved TAVALISSE for the treatment of thrombocytopenia in adult patients with chronic ITP who have had an insufficient response to a previous treatment. In July 2025, Kissei announced that its licensing partner, JW Pharmaceutical Corporation, commercially launched TAVALISSE in Korea.
We have exclusive commercial and license agreements with Medison to commercialize fostamatinib in all potential indications in Canada and Israel. In November 2020, Health Canada approved the New Drug Submission for TAVALISSE for the treatment of thrombocytopenia in adult patients with chronic ITP who have had an insufficient response to other treatments. In August 2021, Medison Israel received the licenses for registrational approval from the Ministry of Health. TAVALISSE is commercially available in Canada and Israel.
We have a commercial license agreement with Knight under which Knight has exclusivity rights to commercialize fostamatinib for approved indications in Latin America, consisting of Mexico, Central and South America, and the Caribbean, and we are responsible for the exclusive manufacture and supply of fostamatinib for all development and commercialization activities under a related supply agreement. Knight submitted Marketing Authorization Applications (MAAs) in Mexico, Colombia, Brazil, Argentina and Paraguay for fostamatinib for the treatment of adult patients with chronic ITP who had insufficient response to a previous treatment. In December 2024, Knight announced that TAVALISSE was approved in Mexico for this indication.
REZLIDHIA/Olutasidenib in R/R AML with mIDH1
REZLIDHIA overview
mIDH1 alterations are seen in AML, MDS, glioma, chondrosarcoma, and intrahepatic cholangiocarcinoma. It is estimated that there are approximately 1,000 adult patients, a well-identified patient population, with mIDH1 R/R AML, part of an AML market estimated to have an incidence of approximately 22,720 cases in the US in 2026, and an estimated 120,000 cases globally. Despite having approved treatment options for R/R AML patients who are mIDH1 positive, an unmet need remains.
Olutasidenib, an oral, small molecule drug designed to selectively bind to and inhibit mIDH1, is a treatment option with durable remissions, reduced QTc potential (referring to a lower observed impact on the heart rate–corrected QT interval on electrocardiogram), and a stable pharmacokinetics profile that enables a consistent drug exposure over time. This targeted agent has the potential to provide therapeutic benefit by reducing 2-hydroxyglutarate levels and restoring normal cellular differentiation. IDH1 is a natural enzyme that is part of the normal metabolism of all cells. When mutated, IDH1 activity can promote blood malignancies and solid tumors. Olutasidenib was granted orphan drug designation by the FDA for the treatment of AML, which provides orphan drug market exclusivity from the time of marketing approval on December 1, 2022.
REZLIDHIA is designed to bind to and inhibit mIDH1 to reduce 2-hydroxyglutarate levels and restore normal cellular differentiation of myeloid cells. REZLIDHIA is a novel, non-intensive monotherapy treatment in the R/R AML setting demonstrating a CR+CRh rate of 35% in patients with over 90% of those responders in complete remission.
We in-licensed REZLIDHIA from Forma pursuant to a license and transition services agreement entered in July 2022, with exclusive, worldwide rights for development, manufacturing and commercialization of REZLIDHIA for any uses, including for the treatment of AML and other malignancies. In accordance with the terms of the license and transition services agreement, we paid an upfront fee of $2.0 million, with the potential to pay up to $67.5 million additional payments upon achievement of specified development and regulatory milestones and up to $165.5 million additional payments upon achievement of certain commercial milestones. In 2022, certain milestones were met which entitled Forma to receive a $17.5 million milestone payments. In addition, subject to the terms and conditions of the license and transition services agreement, Forma would be entitled to tiered royalty payments on net sales of licensed products at percentages ranging from low-teens to mid-thirties, as well as certain portions of our sublicensing revenue, subject to certain standard reductions and offsets.
In December 2022, the FDA approved REZLIDHIA capsules for the treatment of adult patients with R/R AML with IDH1 mutation as detected by an FDA-approved test, and we began the commercialization of REZLIDHIA and made it available to patients. The recommended dosage of REZLIDHIA is 150 mg taken orally twice daily until disease progression or unacceptable toxicity. The FDA approval was based on the New Drug Application (NDA) for olutasidenib for the treatment of mIDH1 R/R AML submitted by Forma, that had a PDUFA action date for the application of February 15, 2023. The NDA was supported with a Phase 2 registrational trial for olutasidenib in mIDH1 R/R AML. Interim results from the Phase 2 registrational trial were reported at the American Society of Clinical Oncology (ASCO) annual meeting in June 2021. The interim results of this trial of 153 patients showed that olutasidenib demonstrated a favorable tolerability profile as a monotherapy in patients with R/R AML who have a susceptible mIDH1, and achieved a complete remission (CR) plus CR with partial hematologic recovery (CRh) rate of 33.3% (30% CR and 3% CRh), the primary efficacy endpoint. While a median duration of CR/CRh was not yet reached, a sensitivity analysis (with a hematopoietic stem cell transplant, as the end of a response) indicated the median duration of CR/CRh was 13.8 months. The overall response rate, comprised CR, CRh, Cri, partial response, and morphologic leukemia-free state (MLFS), was 46% and the median duration of overall response rate (ORR) was 11.7 months. The median overall survival was 10.5 months. For patients with CR/CRh, the median overall survival was not reached, but the estimated 18-month survival was 87%. The most frequently reported treatment emergent adverse events were nausea, constipation, increased white blood cell count, decreased red blood cell count, pyrexia, febrile neutropenia, and fatigue.
In January 2023, we announced that REZLIDHIA has been added by the National Comprehensive Cancer Network (NCCN) to the latest NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) for AML. REZLIDHIA is now included as a recommended targeted therapy for adult patients with R/R AML with IDH1 mutation.
In February 2023, we announced peer-reviewed publication data in Blood Advances, which summarize clinical results from the Phase 2 registrational trial of REZLIDHIA in patients with mIDH1 R/R AML. The published data demonstrate that REZLIDHIA induced durable remissions and transfusion independence with a well-characterized safety profile. The observed efficacy is clinically meaningful and represents a therapeutic advance in this poor prognosis patient population with limited treatment options. REZLIDHIA demonstrated both a high rate of response and an extended median duration of complete response of 28.1 months, which is more than a year longer than what is reported with the standard of care. In June 2023, we announced the second REZLIDHIA publication in Blood Advances, a review article examining the preclinical and clinical development, and the positioning of REZLIDHIA in the mIDH1 AML treatment landscape. The review concluded that the approval of REZLIDHIA is a critical addition to the mIDH1 AML treatment landscape. Further, the available data supports the use of REZLIDHIA as monotherapy in R/R AML patients who have failed intensive chemotherapy or venetoclax plus hypomethylating agents combination therapy.
In April 2024, we announced a peer-reviewed publication in Leukemia & Lymphoma on data from an analysis of the Phase 2 study evaluating REZLIDHIA in patients with mIDH1 AML who are R/R to prior venetoclax-based regimens.
The findings from these analyses suggest that REZLIDHIA alone or in combination with azacitidine demonstrated potential efficacy in patients with AML following failure of venetoclax combination therapy.
In May 2024, we announced the presentation of the registrational Phase 2 trial of REZLIDHIA in R/R mIDH1 AML patients at the 2024 ASCO Annual Meeting and EHA 2024 Hybrid Congress. The data presented reinforces REZLIDHIA’s efficacy in heavily pretreated patients with mIDH1 AML, including those R/R to prior venetoclax. The safety profile was consistent with what was previously reported. Further, REZLIDHIA was generally well tolerated in elderly patients with R/R mIDH1 AML and induced remissions. Despite the challenges of treating elderly patients who had already failed prior AML treatment, the results suggest that elderly patients can benefit from therapy with REZLIDHIA. REZLIDHIA was also effective in achieving remission in patients with mIDH1 R/R AML and served as a bridging strategy towards potentially curative allogeneic transplantation in a substantial subset of these previously ineligible patients. Additionally, REZLIDHIA was well tolerated in a subset of patients with myeloproliferative neoplasms mIDH1 AML, a patient population often associated with poor responses to available therapies.
In October 2025, we announced the publication of the final five-year data for REZLIDHIA in patients with R/R mIDH1 AML in the Journal of Hematology and Oncology. The publication reports the final follow-up analysis of the registrational Phase 2 trial, with an additional two years of efficacy and safety data. These five-year data further support the durable responses and manageable safety profile observed with olutasidenib in patients with R/R mIDH1 AML, including those R/R to prior venetoclax. The safety profile remained consistent with what was previously reported, with no new safety signals identified.
Competitive landscape for REZLIDHIA
There is currently one other product approved in the US for patients with IDH1 mutation. The FDA granted approval to TIBSOVO® (ivosidenib), an oral targeted IDH1 mutation inhibitor, (i) in July 2018, for adult patients with R/R AML with a susceptible IDH1 mutation, (ii) in May 2019, for newly diagnosed AML with a susceptible IDH1 mutation who are at least 75 years old or who have comorbidities that preclude use of intensive induction chemotherapy, (iii) in August 2021, for adult patients with previously treated, locally advanced or metastatic cholangiocarcinoma with an IDH1 mutation as detected by an FDA-approved test, (iv) in May 2022, in combination with azacitidine (azacitidine for injection) for newly diagnosed AML with a susceptible IDH1 mutation, as detected by an FDA-approved test in adults 75 years or older, or who have comorbidities that preclude use of intensive induction chemotherapy, and (v) in October 2023, for adult patients with R/R MDS with a susceptible IDH1 mutation, as detected by an FDA-approved test. In addition, some clinicians may utilize non-targeted treatments for patients with mIDH1 R/R AML, including use of venetoclax combinations, hypomethylating agents, other chemotherapy regimens, or investigational agents that may be available to them.
REZLIDHIA commercial activities, including sales and marketing
We believe REZLIDHIA is highly synergistic with our existing hematology-oncology focused commercial and medical affairs infrastructure. Our commercial effort focuses on growing awareness of REZLIDHIA within key institutions, and among targeted HCPs who manage patients with R/R AML with mIDH1. We retain the global rights, excluding certain geographies as discussed below, to develop and commercialize olutasidenib for all indications, and we are currently exploring other ex-US partnership opportunities.
Olutasidenib outside of the US
We have a collaboration and license agreement with Kissei for an exclusive right to develop and commercialize olutasidenib in all human diseases in Japan, Korea and Taiwan. Kissei will initially seek approval for REZLIDHIA in Japan for R/R mIDH1 AML and will be responsible for conducting clinical studies as required by the Japanese PMDA. We remain responsible for the manufacture and supply of olutasidenib for all development and commercialization activities and will supply Kissei with bulk drug product for use under the license and supply agreements.
We also have a commercial license agreement with Dr. Reddy’s for an exclusive license to develop and commercialize olutasidenib in Dr. Reddy’s territory which includes Latin America, South Africa, India, certain countries in the CIS, Southeast Asia region and North Africa, Australia, and New Zealand. We are responsible for the exclusive manufacture and supply of olutasidenib for all future development and commercialization activities under a supply agreement.
Under the license and services agreement with Forma, Forma is entitled to a certain portion of sublicensing revenue, which include, but are not limited to upfront payment, milestone payments and royalties, that we receive from a third party sublicensee. Following the license agreements with Kissei and Dr. Reddy’s as discussed above, Forma is entitled to a portion of the sublicensing revenue we receive from Kissei and Dr. Reddy’s.
GAVRETO/Pralsetinib in metastatic RET fusion-positive NSCLC and advanced thyroid cancers
GAVRETO overview
RET is a receptor tyrosine kinase that activates multiple downstream pathways involved in cell proliferation and survival. RET can be activated by mutation or when a portion of the RET gene that encodes the kinase domain is joined to part of another gene creating a fusion gene that encodes an aberrantly activated RET fusion protein. RET alterations, such as fusions or mutations, drive the growth of multiple tumor types. It is estimated that over 229,000 adult patients in the US will be diagnosed with lung cancer in 2026. NSCLC is the most common type of lung cancer in the US accounting for 77% of all lung cancer diagnoses. RET activating fusions are key disease drivers in NSCLC. RET fusions are implicated in approximately 1-2% of patients with NSCLC.
We acquired the rights to research, develop, manufacture and commercialize GAVRETO from Blueprint, pursuant to an Asset Purchase Agreement entered in February 2024. GAVRETO is a once daily, small molecule, oral, kinase inhibitor of wild-type RET and oncogenic RET fusions. Currently, GAVRETO is one of only two approved RET inhibitors on the market for patients. GAVRETO is approved by the FDA for the treatment of adult patients with metastatic RET fusion-positive NSCLC as detected by an FDA-approved test.
GAVRETO is also approved for the treatment of adult and pediatric patients 12 years of age and older with advanced or metastatic RET fusion-positive thyroid cancer who require systemic therapy and who are radioactive iodine-refractory (if radioactive iodine is appropriate). This indication was approved by the FDA under accelerated approval based on overall response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial. Discussions with the FDA regarding confirmatory requirements are ongoing.
In June 2024, we announced the completion of the transfer to us of the NDA for GAVRETO, and GAVRETO became commercially available from us in the US by prescription. GAVRETO was co-marketed by Blueprint and Genentech, a member of Roche Group (Roche), to patients in the US since September 2020 pursuant to a collaboration agreement between Blueprint and Roche, which agreement was terminated effective in February 2024.
On December 22, 2025, the FDA notified us of the approval of a Prior Approval supplemental NDA for GAVRETO, which updated the US Prescribing Information to add a boxed warning regarding serious infections, including opportunistic infections. We previously communicated this risk information to healthcare providers via a Dear Healthcare Provider letter in October 2024. The FDA also notified us that we have met our postmarketing commitment for GAVRETO from our September 2020 accelerated approval to submit the final report for the AcceleRET-Lung study.
In January 2026, initial data from the TAPISTRY study was presented in a poster presentation at the ASCO Gastrointestinal Cancers Symposium. TAPISTRY is a Phase 2, global, open-label, multicohort study evaluating the efficacy and safety of pralsetinib in patients with RET fusion-positive solid tumors, including pancreatic, colorectal, and hepatobiliary cancers. The reported analysis included results from an efficacy-evaluable population of 39 patients, in which pralsetinib demonstrated an overall response rate of 67%. These results support continued evaluation of pralsetinib in RET fusion-positive tumors beyond lung cancer; however, further clinical investigation is ongoing, and there can be no assurance that these results will be confirmed in future studies or lead to regulatory approval for additional indications.
In March 2026, we announced the publication of the final data from the registrational trial evaluating pralsetinib for the treatment of patients with metastatic RET fusion-positive NSCLC in the Journal of Clinical Oncology. The final data, which includes an additional 42 months of follow-up from data previously published, further supports the robust, durable responses with a manageable safety profile and no new safety signals identified in treatment-naïve and previously treated patients with RET fusion-positive NSCLC and advanced or metastatic thyroid carcinoma.
The NCCN Guidelines for NSCLC recommends pralsetinib as a preferred first-line treatment option for RET+ patients, including for patients identified during first-line treatment with systemic therapy.
The FDA granted GAVRETO new chemical entity exclusivity until September 2025 and orphan drug exclusivity until September 2027 with respect to the approval for treatment of adult patients with metastatic RET fusion-positive NSCLC as detected by an FDA-approved test. The FDA also granted GAVRETO two orphan drug exclusivities until December 2027 with respect to FDA approval for the treatment of adult and pediatric patients 12 years of age and older with advanced or metastatic RET fusion-positive thyroid cancer who require systemic therapy and who are radioactive iodine-refractory (if radioactive iodine is appropriate), and for the treatment of adult and pediatric patients 12 years of age and older with advanced or metastatic RET-mutant medullary thyroid carcinoma who require systemic therapy.
Competitive landscape for GAVRETO
GAVRETO faces competition for RET fusion-positive NSCLC and advanced thyroid cancers from Lilly’s selpercatinib (Retevmo®). In addition, other commercially available therapies used to treat RET fusion-positive NSCLC include cabozantanib and platinum-based chemotherapy regimens with or without pembrolizumab, atezolizumab, nivolumab/ipilumumab, cemiplimab or tremelimumab-durvalumab. GAVRETO may also face competition from other drug candidates in development for RET-altered cancers, as well as multi-kinase inhibitors with RET activity being evaluated in clinical trials.
GAVRETO commercial activities, including sales and marketing
We began our commercialization and started recognizing revenue from product sales of GAVRETO in June 2024. We believe GAVRETO is highly synergistic with our current product portfolio, and we expect to continue to leverage our existing commercial infrastructure to ensure current and newly prescribed GAVRETO patients have continued access to this important treatment option. We distribute and market GAVRETO for approved indications in RET fusion-positive NSCLC and advanced thyroid cancers.
Clinical Stage Programs
R289, an Oral IRAK1/4 Inhibitor for Hematology-Oncology, Autoimmune, and Inflammatory Diseases
During the second quarter of 2018, we selected R835, a proprietary molecule from our dual IRAK1/4 inhibitor program, for human clinical trials. This investigational candidate is an orally administered, potent and selective inhibitor of IRAK1 and IRAK4 that blocks inflammatory cytokine production in response to toll-like receptor (TLR) and the interleukin-1 receptor (IL-1R) family signaling. TLRs and IL-1Rs play a critical role in the innate immune response and dysregulation of these pathways can lead to a variety of inflammatory conditions. R835 prevents cytokine release in response to TLR and IL-1R activation in vitro, and is active in multiple rodent models of inflammatory disease including psoriasis, arthritis, lupus, multiple sclerosis and gout. Preclinical studies show that R835 inhibits both the IRAK1 and IRAK4 signaling pathways, which play a key role in inflammation and immune responses to tissue damage. Dual inhibition of IRAK1 and IRAK4 allows for more complete suppression of pro-inflammatory cytokine release than inhibition of either one individually.
In October 2019, we announced results from a Phase 1 randomized, placebo-controlled, double-blind clinical study evaluating the safety, tolerability, pharmacokinetics (PK) and pharmacodynamics of R835 in 91 healthy adult subjects. The Phase 1 study showed that R835 had a favorable safety, tolerability, and PK profile and established proof-of-mechanism by demonstrating the inhibition of inflammatory cytokine production in response to a lipopolysaccharide (LPS) challenge.
We advanced the development of our IRAK1/4 inhibitor program, following evaluation of single and multiple ascending doses of R289, a new pro-drug formulation of R835, in healthy subjects. In January 2022, we initiated a Phase 1b open-label, multicenter study to evaluate the safety, tolerability and preliminary efficacy of R289 in patients with R/R lower-risk MDS. In December 2022, we announced the dosing of the first patient. This Phase 1b study is expected to enroll approximately 86 patients (up to 36 patients in the dose escalation phase, up to 40 patients in the dose expansion phase, and 10 patients in an exploratory cohort evaluating post- or ineligible ESA treatment naïve patients). The primary objective of the study is safety, with secondary and exploratory objectives to assess preliminary efficacy and characterize the pharmacokinetic and pharmacodynamic profile of R289. Enrollment in the dose escalation part of the study was completed in July 2025. In October 2025, we announced enrollment of the first patient in the dose expansion part of the study, where up to 40 patients will be randomized to receive either 500 mg once daily or twice daily to determine the recommended Phase 2 dose for future clinical studies. Enrollment is ongoing and we expect to complete enrollment of the dose expansion phase of the Phase 1b study and select the recommended Phase 2 dose for future clinical studies in the second half of 2026. We anticipate sharing preliminary data from the dose expansion phase of the study by the end of 2026.
In December 2024, initial data from the dose escalation part of the Phase 1b study was presented at the 66th ASH Annual Meeting and Exposition. In summary, R289 was generally well tolerated with preliminary signs of efficacy in a heavily pretreated lower-risk MDS patient population, the majority of whom were HTB at baseline. RBC-TI ≥8 weeks was achieved by three patients (1 at 500 mg once daily and 2 at 750 mg once daily); two HTB patients achieved RBC-TI >24 weeks. The median duration of RBC-TI was 29 weeks (range 12.7-51.9 weeks). The three patients that achieved RBC-TI had peak hemoglobin increases exceeding 2.0 g/dL compared to baseline. We also reported that one HTB patient receiving 500 mg once daily achieved a minor HI-E response, with a 64% reduction in RBC transfusions compared to baseline; however, in the July 15, 2025 data cut, we determined that this patient had received blood transfusions that were not captured in the database at the time of the initial data analysis. Accordingly, this patient was subsequently determined to be a non-responder.
In December 2025, we presented the updated data from the dose escalation phase of our ongoing Phase 1b study evaluating R289 in patients with R/R lower-risk MDS, at the 67th ASH Annual Meeting and Exposition. R289 continued to be generally well tolerated in a heavily pretreated R/R lower-risk MDS patient population, the majority of whom were HTB at baseline. As of the October 28, 2025 data cutoff, 33 patients were enrolled in the dose escalation part of the study. Patients had a median age of 75. The median number of prior therapies was 3 (range: 1-8); 76% (25) of patients had received luspatercept, 73% (24) had received an ESA, 67% (22) had received an HMA and 6% (2) had received imetelstat. 61% (20) of patients were HTB at baseline. 67% (22) of patients were ring sideroblast negative. Median duration of treatment was 5.5 months (range: 0.9 - 27.7 months). R289 was generally well tolerated across all dose groups in this heavily pre-treated lower-risk MDS patient population, the majority of whom were HTB at baseline. The most common Grade 1/2 treatment-emergent adverse events were diarrhea, constipation and fatigue, increased creatinine, and cough. The most frequent Grade 3/4 treatment-emergent adverse events were anemia, decreased neutrophil count and pneumonia, and increased ALT and AST. One dose limiting toxicity (Grade 4 AST increased/Grade 3 ALT increased) was reported in the 750 mg dose group. For evaluable transfusion dependent patients (≥16 weeks follow up) at dose levels of at least 500 mg once daily and higher, 6/18 (33%) patients achieved durable RBC-TI of >8 weeks (500 mg once daily (1/3), 750 mg once daily (2/5), 500/250 mg once daily (1/5), 500 mg twice daily (2/5)). Duration of RBC-TI was >16 weeks in 4 patients and >24 weeks in 3 patients. The median time to onset of RBC-TI was 1.9 months and the median duration of RBC-TI was 22.9 weeks. Peak hemoglobin increases of 2.9 to 6.1 g/dL compared to baseline occurred in patients achieving RBC-TI. Of the 6 patients achieving RBC-TI, 5 had received an HMA.
The FDA granted R289 Orphan Drug designation for the treatment of myelodysplastic syndromes in January 2025 and Fast Track designation for the treatment of previously-treated transfusion dependent lower-risk myelodysplastic syndrome in November 2024.
Olutasidenib for mIDH1 AML, Other Hematologic Cancers and HGG
We have a strategic collaboration agreement with MDACC entered in December 2023, to expand our evaluation of olutasidenib in AML and other hematologic cancers with IDH1 mutations. Under such collaboration agreement, we will jointly lead the clinical development efforts with MDACC to evaluate the potential of olutasidenib to treat newly diagnosed and R/R patients with AML, higher-risk MDS, and advanced myeloproliferative neoplasms, in combination with other agents. The collaboration will also support the evaluation of olutasidenib as monotherapy in patients with IDH1 mutated CCUS and lower-risk MDS, as well as maintenance therapy following hematopoietic stem cell transplant. Five studies in the multi-year strategic development alliance are open for enrollment. The five studies include, (i) a Phase 1b/2 triplet therapy trial of decitabine and venetoclax in combination with olutasidenib in patients with mIDH1 AML. The Phase 1b part of the trial seeks to determine the safety and tolerability and recommended Phase 2 dose of decitabine and venetoclax in combination with olutasidenib; (ii) a Phase 2 study in patients with IDH1-mutated CCUS, lower-risk MDS and chronic myelomonocytic leukemia (CMML); (iii) a Phase 1/2 study of olutasidenib maintenance therapy following an allogeneic stem cell transplant for patients with IDH1-mutated myeloid malignancies; (iv) a Phase 2 study of olutasidenib in combination with hypomethylating agents (HMA) in patients with mIDH1 higher-risk myelodysplastic syndrome (HR-MDS)/ CMML or advanced myeloproliferative neoplasms; and (v) a Phase 2 multi-arm, multi-center, open-label, non-randomized clinical study will evaluate olutasidenib in combination with co-targeted therapies in patients with R/R IDH1-mutated myeloid malignancies harboring activated signaling pathway mutations.
In January 2024, we announced our collaboration with CONNECT to conduct a Phase 2 clinical trial to evaluate olutasidenib in combination with temozolomide in patients with HGG harboring an IDH1 mutation. Under the collaboration, CONNECT will include the olutasidenib treatment arm within CONNECT’s TarGet study, a molecularly guided Phase 2 umbrella clinical trial for HGG. In our sponsored arm, TarGet-D, adolescents and young adult patients (ages 12 to 39 years old) with newly-diagnosed IDH1-mutation positive HGG will receive maintenance therapy with olutasidenib in combination with temozolomide for the first year after radiotherapy, followed by olutasidenib monotherapy for the second year. The first patient was enrolled in the Phase 2 TarGet-D study in October 2025.
Partnered Clinical Programs
We have product candidates in clinical development with BerGenBio for the development and commercialization of AXL receptor tyrosine kinase inhibitor, R428 (now referred to as bemcentinib (BGB324)), and with Daiichi to pursue research related to MDM2 inhibitor, DS-3032 (now referred as milademetan). The worldwide rights to milademetan were out-licensed from Daiichi to Rain Oncology Inc., now Pathos AI, Inc.
Research, Preclinical and Clinical Development Programs
We maintain expertise in drug development to leverage our existing proprietary collection of inhibitors, small-molecule compound libraries and large database of associated phenotypic and biochemical assay results of therapeutic interest. We also maintain leading expertise on specific areas of operation such as inhibition of SYK, IRAK1/4 and RIPK1 kinases and mIDH1 to assist clinical development and commercial affairs, as well as to expand and explore additional opportunities for such inhibitors in the clinical space. Our preclinical operations involve collaborations with clinical research organizations, leading investigators from universities and research organizations around the world, and strategic collaborations with other pharmaceutical companies.
We have experts in clinical development to design and implement clinical trials and to analyze the data derived from these trials. The clinical development group possesses expertise in project management and regulatory affairs. We work with external clinical research organizations with expertise in managing clinical trials, drug formulation, and the manufacture of clinical trial supplies to support our clinical development efforts. We also have strategic development collaborations with MDACC and CONNECT to conduct evaluation of olutasidenib in other diseases areas with IDH1 mutations.
Commercialization and Sponsored Research and License Agreements
See “Note 4 – Sponsored Research and License Agreements” and “Note 5 – In-licensing and Acquisition” to our “Notes to Condensed Financial Statements” contained in Part I, Item 1 of this Quarterly Report on Form 10-Q for related discussions.
Results of Operations
Revenues
The following table summarizes revenues for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | Change | | | | | | |
| Product sales, net | $ | 54,923 | | | $ | 43,550 | | | $ | 11,373 | | | | | | | |
| Contract revenues from collaborations | 3,895 | | | 9,783 | | | $ | (5,888) | | | | | | | |
| | | | | | | | | | | |
| Total revenues | $ | 58,818 | | | $ | 53,333 | | | $ | 5,485 | | | | | | | |
The following table summarizes the percentages of revenues from each of our customers who individually accounted for 10% or more of the total net product sales and revenues from collaborations:
| | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | | | |
| McKesson Corporation | 48 | % | | 42 | % | | | | |
| Cencora, Inc. | 23 | % | | 20 | % | | | | |
Optime Care, Inc.^ | — | % | | 11 | % | | | | |
| Cardinal Health, Inc. | 12 | % | | * | | | | |
______________________________________________________________________
* Denotes less than 10%
^ Decrease for Optime Care, Inc. was due to change in distribution channel
Revenue from product sales is related to our sale of our products in the US, net of chargebacks, discounts and fees, government and other rebates and returns. Typically, our first quarter net sales are impacted by the first quarter reimbursement issues such as the resetting of deductibles, co-pays, and other access delays for Medicare patients with plan changes that take effect in January. Consistent with this pattern, our first quarter 2026 net product sales reflected the impact of these seasonal factors, which primarily affected January and February volumes. We observed improving demands through the quarter, with March showing stronger prescription volumes across our product portfolio.
TAVALISSE net product sales for the three months ended March 31, 2026 were $37.3 million, an increase of 31%, compared to $28.5 million for the three months ended March 31, 2025. The increase was primarily driven by higher volumes and higher price per bottle, as well as a favorable impact from lower revenue reserves. REZLIDHIA net product sales for the three months ended March 31, 2026 were $8.0 million, an increase of 31%, compared to $6.1 million for the three months ended March 31, 2025. The increase was primarily driven by higher volumes and higher price per bottle, partially offset by higher revenue reserves. GAVRETO net product sales for the three months ended March 31, 2026 were $9.6 million, an increase of 7%, compared to $9.0 million for the three months ended March 31, 2025. The increase was primarily driven by higher price per bottle and, to a lesser extent, higher volumes
Contract revenues from collaborations for the three months ended March 31, 2026 and 2025 was primarily of revenue from Grifols, Kissei and Medison. Revenue from Grifols was $1.8 million and $4.7 million for the three months ended March 31, 2026 and 2025, respectively, consisting entirely of royalties in 2026 and, in 2025, both royalties and delivery of drug supplies. Revenue from Kissei was $1.8 million and $4.6 million, respectively for the three months ended March 31, 2026 and 2025, respectively, consisting entirely of delivery of drug supplies in 2026 and, in 2025, both delivery of drug supply and a $3.0 million milestone payment recognized in connection with the approval of fostamatinib in Korea. Revenue from Medison was $0.3 million and $0.4 million for the three months ended March 31, 2026 and 2025, respectively, consisting of royalties and delivery of drug supplies.
We expect that revenue from product sales to increase in the coming quarters due to moving past the seasonal reimbursement issues, as well as continued execution of our commercial strategy. However, net product sales may be impacted by the demand from our customers, changes to government and private payor rebate programs, chargeback and discount programs, co-payment assistance programs, and any other rebate and discount programs we may enter in the future. In addition, our future revenues may include payments from our existing and new collaboration partners and government grants. As of March 31, 2026, we had $1.4 million of deferred revenue relating to our collaboration agreement with Kissei which we will recognize as revenue upon satisfaction of our remaining performance obligations.
Cost of Product Sales
The following table summarizes cost of product sales for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | Change | | | | | | |
| Cost of product sales | $ | 4,606 | | | $ | 4,409 | | | $ | 197 | | | | | | | |
The cost of product sales includes the cost of inventories sold to our customers and to our collaborative partners. Certain inventories sold in prior periods were acquired or produced before FDA approval and therefore did not reflect full production costs, as pre-approval manufacturing costs were previously expensed to research and development. Specifically, we utilized zero-cost API inventory for TAVALISSE, which reduced cost of product sales in those periods. As post-approval inventory is acquired or produced, inventory and cost of product sales reflect the full manufacturing cost.
We rely and will continue to rely on certain third parties, including those located outside the US to manufacture our products. The imposition or threat of imposition of trade policies, tariffs (including retaliatory tariffs), taxes and other cross-border operations could result in higher cost of product sales. Cost of product sales may also include reserves for potential excess, dated or obsolete inventories, estimated based upon assumptions about future demand and market conditions as well as product shelf lives. Cost of product sales also includes amortization of intangible assets and royalties.
The increase in cost of product sales for the three months ended March 31, 2026, compared to the same period in 2025, was primarily driven by higher royalties of $0.5 million. This increase was partially offset by a $0.3 million decrease in product costs, primarily due to the timing of drug supply deliveries to collaboration partners, partially offset by higher product costs associated with increased product sales.
Research and Development Expense
The following table summarizes research and development expense for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | Change | | | | | | |
| Research and development expense | $ | 11,676 | | | $ | 8,436 | | | $ | 3,240 | | | | | | | |
| Stock-based compensation expense included in research and development expense | $ | 441 | | | $ | 872 | | | $ | (431) | | | | | | | |
The increase in research and development expense in the three months ended March 31, 2026 compared to the same period in 2025, was primarily due to increased clinical trial related expenses of $2.6 million resulting from the timing of clinical development programs, including the progress activities on our ongoing IRAK1/4 inhibitor program, and increase in other various research and development expenses of $0.7 million .
Our research and development expenditures include costs related to preclinical and clinical trials, scientific personnel, supplies, equipment, consultants, sponsored research, stock-based compensation, and allocated facility costs. We expect to continue to incur significant research and development expense as we continue our activities in our clinical studies including IRAK1/4 inhibitor program; our collaborative partnerships with MDACC and CONNECT to conduct evaluation of olutasidenib in other diseases areas with IDH1 mutations; and any other clinical programs we may pursue in the future.
We do not track fully burdened research and development costs separately for each of our drug candidates. Our research team is focused on identifying and evaluating product candidates in our focused range of therapeutic indications that can be developed into small molecule therapeutics in our own proprietary programs or with potential collaborative partners. “Research” expenses relate primarily to personnel expenses, lab supplies, fees to third-party research consultants and compounds. Our development group leads the implementation of our clinical and regulatory strategies and prioritizes disease indications in which our compounds may be studied in clinical trials. “Development” expenses relate primarily to clinical trials, personnel expenses, costs related to our regulatory filings, lab supplies and fees to third-party research consultants. “Other” expenses primarily consist of allocated facilities costs and allocated stock-based compensation expense relating to personnel in research and development groups.
In addition to reviewing the three categories of research and development expense described in the preceding paragraph, we principally consider qualitative factors in making decisions regarding our research and development programs, which include enrollment in clinical trials and the results thereof, the clinical and commercial potential for our drug candidates and competitive dynamics. We also make our research and development decisions in the context of our overall business strategy, which includes the evaluation of potential collaborations for the development of our drug candidates.
Preclinical testing and clinical development are long, expensive and uncertain processes, and we cannot reliably predict the timing of such clinical trial activities. In general, biopharmaceutical development involves a series of steps, beginning with identification of a potential target and including, among others, proof of concept in animals and Phase 1, 2 and 3 clinical trials in humans. Significant delays in clinical testing could materially impact our product development costs and timing of completion of the clinical trials. We do not know whether planned clinical trials will begin on time, will need to be halted or revamped or will be completed on schedule, or at all. Clinical trials can be delayed for a variety of reasons, including delays in obtaining regulatory approval to commence a trial, delays from scale up, delays in reaching agreement on acceptable clinical trial agreement terms with prospective clinical sites, delays in obtaining institutional review board approval to conduct a clinical trial at a prospective clinical site or delays in recruiting subjects to participate in a clinical trial.
We currently do not have reliable estimates of total costs for a particular drug candidate to reach the market. Our potential products are subject to a lengthy and uncertain regulatory process that may involve unanticipated additional clinical trials and may not result in receipt of the necessary regulatory approvals. Failure to receive the necessary regulatory approvals would prevent us from commercializing the product candidates affected. In addition, clinical trials of our potential products may fail to demonstrate safety and efficacy, which could prevent or significantly delay regulatory approval.
The following table presents our total research and development expense by category (in thousands).
| | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | From January 1, 2007* |
| 2026 | | 2025 | | to March 31, 2026 |
| Categories: | | | | | |
| Research | $ | 30 | | | $ | 733 | | | $ | 271,148 | |
| Development | 11,109 | | | 6,715 | | | 623,651 | |
| Other | 537 | | | 988 | | | 282,333 | |
| $ | 11,676 | | | $ | 8,436 | | | $ | 1,177,132 | |
______________________________________________________________________
*We started tracking research and development expense by category on January 1, 2007.
“Other” expenses in the three months ended March 31, 2026 and 2025 consisted of allocated facilities costs of $0.1 million and $0.1 million, respectively, and stock-based compensation expense of $0.4 million and $0.9 million, respectively.
Selling, General and Administrative Expense
The following table summarizes selling, general and administrative expense for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | Change | | | | | | |
| Selling, general and administrative expense | $ | 30,651 | | | $ | 27,715 | | | $ | 2,936 | | | | | | | |
| Stock-based compensation expense included in selling, general and administrative expense | $ | 3,015 | | | $ | 2,452 | | | $ | 563 | | | | | | | |
The increase in selling, general and administrative expense in the three months ended March 31, 2026 compared to the same period in 2025 was primarily due to a $1.6 million increase in commercial related expenses and a $1.7 million increase in personnel-related costs. These increases were partially offset by decrease in various sales, general and administrative expenses of $0.4 million.
We expect to incur significant selling, general and administrative expenses, and expect our commercial related expenses to increase as we continue to expand our commercial activities.
Interest Income and Interest Expense
The following table summarizes interest income and expense for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | Change | | | | | | |
| Interest income | $ | 1,205 | | | $ | 591 | | | $ | 614 | | | | | | | |
| Interest expense | $ | (1,433) | | | $ | (1,853) | | | $ | (420) | | | | | | | |
Interest income reflects returns earned on our cash and investment holdings, while interest expense relates to borrowing costs on our outstanding term loans with MidCap. The increase in interest income for the three months ended March 31, 2026, compared to the same period in 2025, was primarily driven by higher average investment balances, partially offset by lower interest rates. The decrease in interest expense for the three months ended March 31, 2026, compared to the same period in 2025, was primarily due to scheduled principal payments that reduced our outstanding debt balance, as well as impact of lower interest rates.
Income Taxes
The following table summarizes income tax for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended March 31, | | |
| 2026 | | 2025 | | Change | | | | | | |
| Provision for income taxes | $ | 3,003 | | | $ | 65 | | | $ | 2,938 | | | | | | | |
The quarterly provision for income taxes is determined by applying the estimated annual effective tax rate to the year-to-date pre-tax income, adjusted for any discrete items. The estimated annual effective tax rate is updated at the end of each reporting period.
The provision for income taxes for the three months ended March 31, 2026 primarily consisted of federal income tax expense of $2.4 million and estimated state income taxes of $0.6 million. Prior to the fourth quarter of 2025, we maintained a full valuation allowance against our deferred tax assets. Although we do not expect to incur federal cash income taxes due to sufficient NOL and research and development credit carryforwards, we recognized federal income tax expense based on the estimated impact of utilizing the deferred tax assets associated with such carryforwards. The total tax expense differs from the amount computed at the federal statutory rate primarily due to certain non-deductible expenses and state income taxes.
For the three months ended March 31, 2025, the provision for income taxes primarily consisted of estimated state income taxes. The tax expense differs from the amount computed at the federal statutory rate primarily due to the impact of the valuation allowance and state taxes.
Critical Accounting Policies and Use of Estimates
Our discussion and analysis of our financial condition and results of operations is based upon our financial statements, which have been prepared in accordance with US GAAP. The preparation of these financial statements requires us to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
Our critical accounting estimates and significant accounting policies are described in “Note 1 – Description of Business and Summary of Significant Accounting Policies” to our “Notes to Financial Statements” contained in Part II, Item 8, “Financial Statements and Supplementary Data” of our Annual Report on Form 10-K for the year ended December 31, 2025. There have been no material changes to the accounting policies described in our Annual Report on Form 10-K for the year ended December 31, 2025.
Recent Accounting Pronouncements
See related discussions of recently issued accounting standards in “Note 1 – Organization and Summary of Significant Accounting Policies” to our “Notes to Condensed Financial Statements” contained in Part I, Item 1 of this Quarterly Report on Form 10-Q. We continue to evaluate accounting standards that were recently issued but not yet adopted, as applicable.
Liquidity and Capital Resources
Liquidity
As of March 31, 2026 and December 31, 2025, we had approximately $146.7 million and $155.0 million, respectively, in cash, cash equivalents and short-term investments. We continue to maintain investment portfolios primarily in money market funds, US treasury bills, government-sponsored enterprise securities, corporate bonds and commercial paper. Cash in excess of immediate requirements is invested with a focus to liquidity and capital preservation. We view our investments portfolio as available-for-sale and are available for use in current operations. Wherever possible, we seek to minimize the potential effects of concentration and degrees of risk. We continue to monitor the impact of the changes in the
conditions of the credit and financial markets on our investment portfolio and assess if future changes in our investment strategy are necessary.
The following table summarizes our cash flow activity for the periods presented (in thousands):
| | | | | | | | | | | |
| Three Months Ended March 31, |
| 2026 | | 2025 |
| Net cash provided by (used in): | | | |
| Operating activities | $ | 2,725 | | | $ | (893) | |
| Investing activities | (7,975) | | | (10,552) | |
| Financing activities | (10,919) | | | 484 | |
| Net decrease in cash, cash equivalents and restricted cash | $ | (16,169) | | | $ | (10,961) | |
Net cash provided by operating activities for the three months ended March 31, 2026 reflected net income, adjusted for non-cash items, partially offset by net cash outflows from changes in working capital. These outflows were primarily driven by decreases in liabilities due to the timing of payments, increases in prepaid and other current assets mainly due to the timing of advance payments to contract manufacturers and higher inventory levels due to the timing of production build-up, partially offset by lower accounts receivable due to the timing of collections. In comparison, net cash used in operating activities for the three months ended March 31, 2025 reflected net income, adjusted for non-cash items, offset by net cash outflows from changes in working capital. These outflows were primarily driven by similar factors, including increases in prepaid and other current assets and decreases in liabilities, partially offset by lower inventory levels.
Net cash used in investing activities for the three months ended March 31, 2026 and 2025 consisted of net purchases of short-term investments of $8.0 million and $10.6 million, respectively.
Net cash used in financing activities for the three months ended March 31, 2026 consisted primarily of $7.5 million of principal payments of term loans and $3.7 million of repurchases of common stock in connection with employee tax withholding on RSU vesting, partially offset by $0.3 million of net proceeds from the issuance of common stock under equity plans. Net cash provided by financing activities for the three months ended March 31, 2025 consisted of $0.5 million of net proceeds from issuance of common stock under equity plans.
We believe that our existing capital resources will be sufficient to support our current and projected funding requirements, including the continued commercialization of our products, through at least the next 12 months from this Form 10-Q filing date. We have based this estimate on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we currently expect. Because of the numerous risks and uncertainties associated with commercializing a product, the development of our product candidates and other research and development activities, we are unable to estimate with certainty our future product revenues, our revenues from our current and future collaborative partners, the amounts of increased capital outlays and operating expenditures associated with our current and anticipated clinical trials and other research and development activities.
Capital Resources
We finance our operations primarily through sales of our products, and contract payments under our collaboration agreements, as well as through equity securities and debt financing.
Under our existing collaboration agreements entered in the ordinary course of business, we have received, and may in the future receive upfront cash payments, payments contingent upon the achievement of specified events by our partners, and royalties on net sales of products sold by such partners under the agreements. The total potential future contingent payments due to us under these agreements are approximately $657.1 million. This amount accounts for terminated programs and Lilly’s April 2026 notice to terminate the Lilly Agreement and assumes achievement of all applicable milestones under the existing agreements. The estimate excludes any potential royalties that may be payable to us if our partners successfully commercialize licensed products. Potential future milestone payments under these agreements are contingent solely upon our partners’ future efforts and the achievement of specified development, regulatory, and commercial milestones. See further discussion in “Note 4 – Sponsored Research and License Agreements” to our “Notes to Condensed Financial Statements” contained in Part I, Item 1 of this Quarterly Report on Form 10-Q.
We have an Open Market Sale Agreement with Jefferies LLC (Jefferies), as a sole agent, entered on August 4, 2020, and amended and restated on August 2, 2024. Pursuant to such Open Market Sale Agreement, we may sell from time to time, through Jefferies, shares of our common stock in sales deemed to be “at-the-market offerings” as defined in Rule 415 under the Securities Act, subject to conditions specified in the Open Market Sale Agreement, including maintaining an effective registration statement covering the sale of shares under the Open Market Sale Agreement. We have an active Registration Statement filed with the SEC, which registered, among other securities, a base prospectus which covers the offering, issuance, and sale by us of up to $250.0 million in the aggregate of the securities identified from time to time in one or more offerings, which include the $100.0 million of shares of our common stock that may be offered, issued and sold under the Open Market Sale Agreement. As of March 31, 2026, we have not sold any shares of common stock under such Open Market Sale Agreement.
We have a Credit Agreement with MidCap that provided for $60.0 million term loan credit facility, under which $45.0 million was outstanding as of March 31, 2026. On May 5, 2026, we terminated the Credit Agreement and repaid all outstanding borrowings thereunder, including applicable prepayment premiums, accrued interest and final payment fees, using cash on hand. Concurrently, we entered into a new Credit Agreement with MidCap, which provides for a revolving credit facility with a maximum borrowing capacity of $40.0 million, with an option to increase to $60.0 million, subject to customary conditions. Availability under the revolving credit facility is subject to a borrowing base based primarily on eligible accounts receivable and inventory. While the revolving credit facility enhances our financial flexibility to support operations and working capital needs, our liquidity is dependent on the level of borrowing base availability. As of the date of this filing, we had an outstanding borrowing of $8.0 million under the revolving credit facility.
We may from time to time consider raising additional funds through public and/or private offerings of equity securities, debt financings, or from other sources, in order to fund ongoing operations, to strengthen our long-term financial profile or to pursue opportunistic corporate development activities. However, certain external factors such as global geopolitical tensions, political and economic legislations, lingering economic effects of the global pandemic, and other factors may continue to rapidly evolve which could significantly disrupt the global financial markets. Our ability to raise additional funds may be adversely impacted by potential worsening of global economic conditions and volatility in the credit and financial markets in the US and worldwide. We could experience an inability to access additional funds, which could in the future negatively affect our capacity for certain corporate development transactions or our ability to make important, opportunistic investments. To the extent that we raise additional funds through the sale of equity, our shareholders’ ownership interest may experience substantial dilution. Our current credit facility with MidCap and any debt financing that we can obtain in the future may involve operating covenants that may restrict our business. To the extent that we raise additional funds through collaboration and licensing arrangements, we may be required to relinquish some of our rights to our technologies or product candidates or grant licenses on terms that are not favorable to us.
Our future funding requirements will depend upon many factors, including, but not limited to:
•the ongoing costs to commercialize our products, or any other future product candidates, if any such candidate receives regulatory approval for commercial sale;
•our ability to generate expected revenue from our commercialization efforts;
•the progress and success of our clinical trials and preclinical activities (including studies and manufacture of materials) of our product candidates conducted by us;
•our ability to secure and maintain our patent protection and regulatory rights;
•our ability to meet operating covenants under our current and future credit facilities, if any;
•our ability to enter into partnering opportunities across our pipeline within and outside the US;
•the costs and timing of regulatory filings and approvals by us and our collaborators;
•the progress of research and development programs carried out by us and our collaborative partners;
•any changes in the breadth of our research and development programs;
•the ability to achieve the events identified in our collaborative agreements that may trigger payments to us from our collaboration partners;
•our ability to acquire or license other technologies or compounds that we may seek to pursue;
•our ability to manage our growth;
•competing technological and market developments;
•the costs and timing of obtaining, enforcing and defending our patent and other intellectual property rights, including regulatory rights such as regulatory data exclusivities;
•expenses associated with any unforeseen litigation, including any arbitration and securities class action lawsuits; and
•pressures on and uncertainty surrounding the US federal government policies, and potential changes in budgetary priorities.
Insufficient funds may require us to delay, scale back or eliminate some or all of our commercial efforts and/or research or development programs, to lose rights under existing licenses or to relinquish greater or all rights to product candidates at an earlier stage of development or on less favorable terms than we would otherwise choose or may adversely affect our ability to operate as a going concern.
Material Cash Requirements
We conduct our commercial activities and research and development programs internally and with third parties that include, among others, arrangements with vendors, consultants, contract research organizations (CROs) and universities. Our contract arrangements with these third parties are generally cancellable on reasonable notice, and our obligations under such arrangements are generally based on services performed. We have agreements with certain clinical research organizations to conduct our clinical trials including our strategic development collaborations with MDACC and CONNECT. The timing of payments for any amounts owed under the respective agreements depends on various factors including, but not limited to, patient enrollment and other progress of the clinical trials. We can terminate these agreements at any time, and if terminated, we would not be liable for the full amount of the respective agreements. Instead, we will be liable for services provided through the termination date plus certain cancellation charges, if any, as defined in each of the respective agreements. In addition, these agreements may, from time to time, be subjected to amendments as a result of any change orders executed by the parties. We expect to continue entering into contracts in the normal course of business with various third parties to support our commercial activities and research and development programs.
In the ordinary course of business, we enter into agreements with contract manufacturers to manufacture our inventory products. These agreements generally include termination provisions that may require us to pay cancellation fees, which vary depending on the timing of termination and may equal up to the full value of the work order. In October 2024, we entered into an agreement with a third-party contract manufacturer to manufacture TAVALISSE, with deliveries expected from 2026 through 2029. As of March 31, 2026, the contractual obligation not included in our financial statements related to an agreement that may potentially be subjected to cancellation fees were approximately $19.2 million, of which, $5.4 million is expected to be due in the remainder of 2026, and $9.5 million is expected to be due in 2027 and 2028. As of March 31, 2026, we have not incurred any cancellation fees under our agreements with contract manufacturers.
Also, as discussed in detail in “Note 5 – In-licensing and Acquisition” of our “Notes to Condensed Financial Statements” contained in Part I, Item 1 of this Quarterly Report on Form 10-Q, pursuant to our license and transition services agreement with Forma, Forma is entitled to potential development and regulatory milestone payments and tiered royalty payments on net sales as well as certain portion of sublicensing revenue. Further, following our olutasidenib sublicensing agreements with Kissei and Dr. Reddy’s, Forma is entitled to a portion of the sublicensing revenue we receive from Kissei and Dr. Reddy’s under such respective agreements.
Additionally, as discussed in detail in “Note 5 – In-licensing and Acquisition” of our “Notes to Condensed Financial Statements” contained in Part I, Item 1 of this Quarterly Report on Form 10-Q, pursuant to an Asset Purchase Agreement with Blueprint, in addition to unpaid purchase price consideration, Blueprint is entitled to potential commercial and regulatory milestone payments, as well as tiered royalty payments.
As discussed above, in connection with the termination of our Credit Agreement with MidCap and entry into the new Credit Agreement, we repaid all outstanding amounts under the term loan facility on May 5, 2026, which was scheduled to mature on September 1, 2027 and had an outstanding principal balance of $45.0 million as of March 31, 2026; such repayment included all outstanding principal, together with applicable prepayment premiums, accrued interest and final payment fees. The revolving credit facility under the new Credit Agreement has a five-year term and bears interest at a rate equal to one-month SOFR, subject to a 2.00% floor, plus an applicable margin of 4.00%. The obligations under the revolving facility is secured by substantially all of our assets, including our intellectual property.
As of March 31, 2026, we have a contractual commitment related to our leased facility, which lease will expire in July 2027. As of March 31, 2026, our contractual commitment related to the lease agreements was $0.9 million, of which $0.7 million is payable in the next 12 months.
We are also subject to claims related to the patent protection of certain of our technologies, other litigations, and other contractual agreements. We are required to assess the likelihood of any adverse judgments or outcomes to these matters as well as potential ranges of probable losses. A determination of the amount of reserves required, if any, for these contingencies is made after careful analysis of each individual matter. We do not have other material contractual commitments with respect to matters discussed above.
Item 3. Quantitative and Qualitative Disclosures About Market Risk
We are exposed to market risks in the ordinary course of our business. These risks primarily include interest rate sensitivities related to our investments and borrowings. There were no material changes to our quantitative and qualitative disclosures about market risks during the three months ended March 31, 2026 as disclosed in “Item 7A. Quantitative and Qualitative Disclosures About Market Risks” of our Annual Report on Form 10-K for the year ended December 31, 2025.
Item 4. Controls and Procedures
Evaluation of Disclosure Controls and Procedures. Based on the evaluation of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act), our chief executive officer (who serves as our principal executive officer) and our chief financial officer (who serves as our principal financial officer) have concluded that, as of the end of the period covered by this report, our disclosure controls and procedures were effective.
Changes in Internal Controls. There were no changes in our internal control over financial reporting that occurred during the quarter ended March 31, 2026 that have materially affected or are reasonably likely to materially affect our internal control over financial reporting.
Limitations on the Effectiveness of Controls. A control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the controls are met. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues, if any, within a company have been detected. Accordingly, our disclosure controls and procedures are designed to provide reasonable, not absolute, assurance that the objectives of our disclosure control system are met and, as set forth above, our chief executive officer and chief financial officer have concluded, based on their evaluation as of the end of the period covered by this report, that our disclosure controls and procedures were sufficiently effective to provide reasonable assurance that the objectives of our disclosure control system were met.
PART II. OTHER INFORMATION
Item 1. Legal Proceedings
From time to time, we may be a party or subject to legal proceedings and claims, either asserted or unasserted, which arise in the ordinary course of business. Some of these proceedings that we may be involved in the future, are claims that are subject to substantial uncertainties and unascertainable damages or other remedies.
Our threshold for disclosing material environmental legal proceedings involving a government authority where potential monetary sanctions are involved is $1.0 million.
Item 1A. Risk Factors
In evaluating our business, you should carefully consider the following risks, as well as the other information contained in this Quarterly Report on Form 10-Q. These risk factors could cause our actual results to differ materially from those contained in forward-looking statements we have made in this Quarterly Report on Form 10-Q and those we may make from time to time. If any of the following risks actually occurs, our business, financial condition and operating results could be harmed. The risks and uncertainties described below are not the only ones facing us. Additional risks and uncertainties not presently known to us, or that we currently see as immaterial, may also harm our business.
We have marked with an asterisk (*) those risk factors below that reflect a substantive change from the risk factors included in our Annual Report on Form 10-K for the year ended December 31, 2025 filed with the SEC on March 3, 2026, if any.
Risk Factor Summary
•Our prospects are highly dependent on our existing commercial products. To the extent that the commercial success of our products in the US and respective territories outside of the US is diminished or halted, our business, financial condition and results of operations may be adversely affected, and the price of our common stock may decline.
•We may not be able to successfully develop or commercialize our product candidates if problems arise in the clinical testing and/or approval process. There is a high risk that drug discovery and development efforts might not generate successful product candidates. If the results of our clinical trials do not meet the primary efficacy endpoints, or if the top-line data from the results of our clinical trials may not ultimately meet the requirements for an NDA approval by the FDA and other regulatory authorities, the commercial prospects of our business may be harmed, and our ability to generate product revenues may be delayed or eliminated.
•Our strategy to expand our hematology and oncology pipeline on our own, or through acquisitions or in-licensing of early or late-stage products or companies, or through partnerships with pharmaceutical and biotechnology companies, as well as academic institutions and government organizations, may not be successful.
•Even if we, or any of our collaborative partners, are able to continue to commercialize our products or any product candidate that we, or they, develop, the product may become subject to unfavorable pricing regulations, unfavorable health technology assessments (HTA), third-party payor reimbursement practices or labeling restrictions, all of which may vary from country to country and any of which could harm our business.
•If we are unable to successfully market and distribute our products and retain experienced commercial personnel, our business will be substantially harmed.
•We are subject to stringent and evolving healthcare regulatory, privacy and information security laws, regulations, rules, policies and contractual obligations, and changes in such laws, regulations, rules, policies, contractual obligations and our actual or perceived failure to comply with such requirements could subject us to significant investigations, audits, fines, penalties, and claims, any of which may have a material adverse effect on our business, financial condition, results of operations or prospects.
•If manufacturers obtain approval for generic versions of our products, or of products with which we compete, our business may be harmed.
•Unforeseen safety issues could emerge with our products that could require us to change the prescribing information, including to add new or more significant warnings (including boxed warnings, the FDA's most prominent safety warning), limit use of the product, and/or result in litigation. Any of these events could have a negative impact on our business.
•We rely and may continue to rely on third-party distribution facilities for the sale of our products and potential sale of any of our product candidates. If any or all of them become subject to adverse findings from inspections or face other difficulties to operate, then the distribution of our products may be interrupted or otherwise adversely affected.
•We lack the capability to manufacture compounds for clinical development and we intend to rely on third parties for commercial supply, manufacturing and distribution, if any, of our product candidates which receive regulatory approval and we may be unable to obtain required material or product in a timely manner, at an acceptable cost or at a quality level required to receive regulatory approval.
•Any product for which we have obtained regulatory approval, or for which we obtain approval in the future, is subject to, or will be subject to, extensive ongoing regulatory requirements by the FDA, EMA, MHRA and other comparable regulatory authorities, and if we fail to comply with regulatory requirements or if we experience unanticipated problems with our products, we may be subject to penalties, we may be unable to generate revenue from the sale of such products, our potential for generating positive cash flow may be diminished, and the capital necessary to fund our operations will be increased. Additionally, approval of a drug under the accelerated drug approval program may be withdrawn or the labeled indication of the drug changed if trials fail to verify clinical benefit or do not demonstrate sufficient clinical benefit to justify the risks associated with the drug.
•If our corporate collaborations or license agreements are unsuccessful, or if we fail to form new corporate collaborations or license agreements, our research and development efforts could be delayed.
•Our success is dependent on securing intellectual property rights and data exclusivity and other regulatory rights (such as orphan exclusivity, pediatric extensions and supplementary protection certificate) held by us and third parties, and our interest in such rights is complex and uncertain.
•If a dispute arises regarding the infringement or misappropriation of the proprietary rights of others, such dispute could be costly and result in delays in our research and development activities, partnering and commercialization activities.
•If our competitors develop technologies that are more effective than ours, our commercial opportunity will be reduced or eliminated.
•If product liability lawsuits are successfully brought against us, we may incur substantial liabilities and may be required to limit commercialization of our products.
Risks Related to Our Business and Our Industry
If the market opportunities for our products and product candidates are smaller than we believe they are, our revenues may be adversely affected, and our business may suffer.
Certain of the diseases that our products and our other product candidates being developed to address are in underserved and underdiagnosed populations. Our projections of both the number of people who have these diseases, as well as the subset of people with these diseases who will seek treatment utilizing our products or product candidates, may not be accurate. If our estimates of the prevalence or number of patients potentially on therapy prove to be inaccurate, the market opportunities for our products and our other product candidates may be smaller than what we believe they are, our prospects for generating expected revenue may be adversely affected and our business may suffer.
We may need to continue to increase the size of our organization and we may encounter difficulties with managing our growth, which could adversely affect our business and results of operations.
We may need to add additional qualified personnel and resources to support our commercial activities and expected growth. Our current infrastructure may be inadequate to support our development and commercialization efforts and expected growth. Future growth will impose significant added responsibilities on members of management, including the need to identify, recruit, maintain and integrate additional employees, and may take time away from running other aspects of our business, including commercialization of our products and development of our other product candidates.
Our future financial performance and our ability to sustain successful commercialization of our products and our ability to commercialize other product candidates that may receive regulatory approval will depend, in part, on our ability to manage any future growth effectively. In particular, as we continue to commercialize our products, we will need to support the training and ongoing activities of our sales force and will likely need to continue to expand the size of our employee base for managerial, operational, financial and other resources. To that end, we must be able to successfully:
•manage our development efforts effectively;
•integrate additional management, administrative and manufacturing personnel;
•further develop our marketing and sales organization; and
•maintain sufficient administrative, accounting and management information systems and controls.
We may not be able to accomplish these tasks or successfully manage our operations and, accordingly, may not achieve our research, development, and commercialization goals. Our failure to accomplish any of these goals, including as a result of business or other interruptions resulting from a potential pandemic or global economic slowdown, could adversely affect our business and operations.
Our strategy to expand our hematology and oncology pipeline on our own, or through acquisitions or in-licensing of early or late-stage products or companies, or through partnerships with pharmaceutical and biotechnology companies, as well as academic institutions and government organizations, may not be successful.
Our business is focused on the development and commercialization of novel therapies that significantly improve the lives of patients with hematologic disorders and cancer. In this regard, we continue to pursue internal drug discovery efforts or partnerships with pharmaceutical and biotech companies, as well as academic institutions and government organizations, with the goal of identifying new product candidates to advance into clinical trials. Our discovery efforts to identify new product candidates require substantial technical, financial and human resources. These discovery efforts may initially show promise in identifying potential product candidates, yet ultimately fail to yield product candidates for clinical development for a number of reasons. For example, potential product candidates may, on later stage clinical trial, be shown to have inadequate efficacy, harmful side effects, suboptimal pharmaceutical profiles or other characteristics suggesting that they are unlikely to be commercially viable products.
Apart from our discovery efforts, we continue to seek to broaden and diversify our product portfolio through acquisition or in-licensing of a product. This strategy is dependent on our ability to successfully identify and acquire or in-license relevant product candidates. In July 2022, we entered into a license and transition services agreement with Forma for an exclusive license to develop, manufacture and commercialize olutasidenib, a proprietary inhibitor of mIDH1, for any uses worldwide, including for the treatment of AML and other malignancies. In December 2022, the FDA approved REZLIDHIA capsules for the treatment of adult patients with R/R AML with a susceptible IDH1 mutations as detected by an FDA-approved test. REZLIDHIA is our second commercial product and we believe is highly synergistic with our existing hematology-oncology focused commercial and medical affairs infrastructure. Further, in February 2024, we entered into an Asset Purchase Agreement with Blueprint to purchase certain assets comprising the right to research, develop, manufacture and commercialize GAVRETO, Blueprint’s proprietary RET inhibitor of tyrosine kinase for the treatment of metastatic RET fusion-positive NSCLC and advanced thyroid cancer, in the US. Simultaneously and in connection with entering into the asset purchase agreement, we also entered into certain supporting agreements with Blueprint, including a customary transition agreement, pursuant to which, during a transition period, Blueprint will transition regulatory and distribution responsibility for pralsetinib to us. In June 2024, we announced the completion of the transfer of GAVRETO NDA to us, and GAVRETO became commercially available from us in the US by prescription. The in-licensing and acquisition of a product is a highly competitive area, and many other companies are pursuing the same or similar product candidates to those that we may consider attractive. In particular, larger companies with more well-established and diverse revenue streams may have a competitive advantage over us due to their size, financial resources and more extensive clinical development and commercialization capabilities. Furthermore, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. The success of this strategy depends partly upon our ability to identify, select and acquire or in-license promising product candidates and technologies. The process of proposing, negotiating and implementing a license or acquisition of a product candidate is lengthy and complex, and we may be unable to in-license or acquire the rights to any such products, product candidates or technologies from third parties for several reasons. We may also be unable to in-license or acquire additional relevant product candidates on acceptable terms. Further, even if we identify acquisition or in-licensing targets, we may not be able to complete the transactions or we may determine after due diligence investigation not to pursue identified targets. Even if we succeed in our efforts to obtain rights to suitable product candidates, the success of our investments in these areas, our investment strategy will remain subject to the inherent risks associated with the development and commercialization of the product, and with the competitive business environment in which we operate.
In addition, acquisitions and in-licensing may entail numerous operational, financial and legal risks, including:
•potential failure of the due diligence process to identify significant problems, liabilities or other shortcomings or challenges of an acquired or licensed product candidate or technology, including problems, liabilities or
other shortcomings or challenges with respect to intellectual property, product quality, partner disputes or issues and other legal and financial contingencies and known and unknown liabilities;
•inability to integrate the target company or in-licensed asset successfully into our existing business and inability to maintain the key business relationships of the target;
•in an in-licensing or an asset acquisition of a product that is commercially available in the market, we may not be able to successfully transition the existing patients who are dependent to the acquired or in-licensed product, or successfully enter into a reimbursement coverage contracts that the existing patients were previously dependent into, or successfully enter into a contract with contract manufacturers to continue the production of the in-licensed or acquired product;
•assumption of unknown or contingent liabilities or incurrence of unanticipated expenses;
•exposure to known and unknown liabilities, including possible intellectual property infringement claims, violations of laws, tax liabilities and commercial disputes;
•incurrence of substantial debt, dilutive issuances of securities or depletion of cash to pay for acquisitions;
•incurrence of large one-time expenses and acquiring intangible assets that could result in significant future amortization expense and significant write-offs;
•higher than expected acquisition and integration costs; and
•inability to maintain uniform standards, controls, procedures and policies.
There is a high risk that drug discovery and development efforts might not generate successful product candidates.
We currently have product candidates in the clinical development and testing stage and may further pursue to expand our development and clinical testing efforts. In our industry, it is statistically unlikely that the limited number of compounds that we have identified as potential product candidates will actually lead to successful product development efforts. We have invested a significant portion of our efforts and financial resources into clinical development. Our ability to generate product revenue, which will not occur until after regulatory approval, if ever, will depend on the successful development, regulatory approval and eventual commercialization of our product candidates.
Our compounds in clinical trials and our future leads for potential drug compounds are subject to the risks and failures inherent in the development of pharmaceutical products. These risks include, but are not limited to, the inherent difficulty in selecting the right drug and drug target and avoiding unwanted side effects, as well as unanticipated problems relating to product development, testing, enrollment, obtaining regulatory approvals, obtaining and maintaining reimbursement in national markets and positive recommendation from HTA bodies, maintaining regulatory compliance, manufacturing, competition and costs and expenses that may exceed current estimates. In future clinical trials, we, our partners or others may discover additional side effects and/or a higher frequency of side effects than those observed in previously completed clinical trials. The results of preliminary and mid-stage clinical trials do not necessarily predict clinical or commercial success, and larger later-stage clinical trials may fail to confirm the results observed in the previous clinical trials. Similarly, a clinical trial may show that a product candidate is safe and effective for certain patient populations in a particular indication, but other clinical trials may fail to confirm those results in a subset of that population or in a different patient population, which may limit the potential market for that product candidate. For example, in October 2024, we issued a Dear Healthcare Provider Letter for GAVRETO related to a new safety signal identified in an ongoing Phase 3 clinical trial of pralsetinib in first-line treatment of RET fusion-positive, metastatic NSCLC patients, being conducted by Roche. The letter advises healthcare providers to apply certain measures to protect patient safety, including enhanced ongoing monitoring for signs and symptoms of infection as well as guidance for withholding treatment to patients in the presence of active infection. On December 22, 2025, the FDA notified us of the approval of a Prior Approval supplemental NDA for GAVRETO, which updated the US Prescribing Information to add a boxed warning regarding serious infections, including opportunistic infections. With respect to our own compounds in development, we have established anticipated timelines with respect to the initiation of clinical trials based on existing knowledge of the compounds. However, we cannot provide assurance that we will meet any of these timelines for clinical development. Additionally, the initial results of a completed earlier clinical trial of a product candidate do not necessarily predict final results and the results may not be repeated in later clinical trials.
Because of the uncertainty of whether the preclinical evidence (pharmacokinetic, pharmacodynamic, safety and toxicity, and/or other factors) or early clinical results will be observed in later clinical trials, we can make no assurances regarding the success of our clinical trials. The impact of those preclinical and clinical results may require us to conduct additional studies, delay, limit or modify clinical trials, or result in more restrictive labeling or other regulatory actions. For example, we conducted a Phase 3 pivotal trial of fostamatinib in patients with warm auto immune hemolytic anemia (wAIHA) initiated in March 2019 and completed in April 2022. In June 2022, we announced top-line efficacy and safety data results of the trial, and the results did not demonstrate statistical significance in the primary efficacy endpoint of durable hemoglobin response in the overall study population. Based on the result of the trial and the guidance from the FDA, we did not file an Supplemental New Drug Application (sNDA) for this indication. Further, we may experience errors, data capture discrepancies at initial data analysis and final study results, or other technical issues in the analysis of our clinical trial results. For example, we conducted our Phase 3 clinical trial to evaluate safety and efficacy of fostamatinib in hospitalized COVID-19 patients launched in November 2020 and completed enrollment in July 2022. We announced in November 2022 that the top-line results did not meet statistical significance in the primary efficacy endpoint. Upon further analysis, we discovered an error by the biostatistical CRO in the application of a statistical stratification factor. After correcting for this statistical error, the primary endpoint of the study was met. However, given the end of the federal COVID-19 PHE in May 2023, and based on feedback from the FDA, DOD and other advisors regarding the program’s regulatory requirements, costs, timeline and potential for success, we decided not to submit an Emergency Use Authorization (EUA) or sNDA. In addition, in December 2024, we presented initial data from the dose escalation part of the Phase 1b study evaluating the safety, tolerability and preliminary efficacy of R289 in patients with R/R lower-risk MDS. We reported that one HTB patient receiving 500 mg once daily achieved a minor HI-E response, with a 64% reduction in RBC transfusions compared to baseline; however, in the July 15, 2025 data cut, we determined that this patient had received blood transfusions that were not captured in the database at the time of the initial data analysis. Accordingly, this patient was subsequently determined to be a non-responder. As the study is ongoing, interim results represent information at the time of the data cut, and final study results will be available after the database lock at the end of the study.
Foreign regulatory requirements governing clinical trials may diverge and impose additional regulatory burdens, which may result in delays. For instance, the new EU Clinical Trials Regulation No 536/2014 (CTR) has amended the system of approval for clinical trials in the EU and has established a new clinical trials portal and database for application for authorizations, called the Clinical Trials Information System (CTIS). All ongoing clinical trials in the EU will be subject to the provisions of the CTR as of January 31, 2025. In addition, on June 18, 2024, new CTIS transparency rules came into effect, requiring scheduled publication of certain key clinical trial information.
If the results of our clinical trials fail to meet the primary efficacy endpoints, or otherwise do not ultimately meet the requirements for an NDA approval by the FDA, the commercial prospects of our business may be harmed, our ability to generate product revenues may be delayed or eliminated or we may be forced to undertake other strategic alternatives that are in our shareholders’ best interests, including cost reduction measures. If we are unable to obtain adequate financing or engage in a strategic transaction on commercially reasonable terms or at all, we may be required to implement further cost reduction strategies which could significantly impact activities related to our commercial efforts and/or research and development of our future product candidates, and could significantly harm our business, financial condition and results of operations. In addition, these cost reduction strategies could cause us to further curtail our operations or take other actions that would adversely impact our shareholders.
We are subject to federal and state healthcare fraud and abuse laws, false claims laws and other federal and state healthcare laws, and the failure to comply with such laws could result in substantial penalties. Our employees, independent contractors, consultants, principal investigators, CROs, commercial partners and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
Our business operations and current and future arrangements with investigators, healthcare professionals, consultants, third-party payors, vendors and customers, may expose us to broadly applicable federal, state and foreign fraud and abuse and other healthcare laws and regulations including anti-kickback and false claims laws, data privacy and security laws, and transparency reporting laws. These laws may constrain the business or financial arrangements and relationships through which we conduct our operations, including how we research, market, sell and distribute any product for which we have obtained regulatory approval, or for which we may obtain regulatory approval in the future. In particular, the promotion, sales and marketing of healthcare items and services, as well as certain business arrangements in the healthcare industry, are subject to extensive laws and regulations intended to prevent fraud, misconduct, bribery kickbacks, self-dealing and other abusive or inappropriate practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, including promoting off-label uses of our products, certain commission compensation, certain customer incentive programs, certain patient support offerings, and other business arrangements generally. Activities subject to these laws also involve the improper use or misrepresentation of information
obtained in the course of patient recruitment for clinical trials, creating fraudulent data in our preclinical studies or clinical trials or illegal misappropriation of drug product, which could result in regulatory sanctions and cause serious harm to our reputation. See “Business – Government Regulation – Healthcare and Privacy Law and Regulation and Healthcare Reform” contained in Part I, Item 1 of our Annual Report on Form 10-K for the year ended December 31, 2025, for more information on the healthcare laws and regulations that may affect our ability to operate.
We are also exposed to the risk of fraud, misconduct or other illegal activity by our employees, independent contractors, consultants, principal investigators, CROs, commercial partners and vendors. Misconduct by these parties could include intentional, reckless and/or negligent conduct that fails to: comply with the laws of the FDA and other similar foreign regulatory bodies; provide true, complete and accurate information to the FDA and other similar foreign regulatory bodies; comply with manufacturing standards we have established; comply with federal and state data privacy, security, fraud and abuse and other healthcare laws and regulations in the US and similar foreign fraudulent misconduct laws; or report financial information or data accurately or to disclose unauthorized activities to us. It is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent inappropriate conduct may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations.
We are also subject to the risk that a person or government could allege such fraud or other misconduct, even if none occurred. Efforts to ensure that our business arrangements will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental and enforcement authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law interpreting applicable fraud and abuse or other healthcare laws and regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant civil, criminal and administrative penalties, damages, disgorgement, monetary fines, imprisonment, additional reporting obligations and oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, contractual damages, reputational harm, diminished profits and future earnings, and curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
We are subject to stringent and evolving privacy and information security laws, regulations, rules, policies, and contractual obligations, and changes in such laws, regulations, rules, policies, contractual obligations and our actual or perceived failure to comply with such requirements could subject us to significant investigations, fines, penalties and claims, any of which may have a material adverse effect on our business, financial condition, results of operations or prospects.*
We collect, use, store, process and transfer personal information and other sensitive data in connection with our clinical trials, research activities and business operations. As a result, we are subject to a wide range of federal, state and foreign laws and regulations relating to privacy, data protection, cybersecurity and, increasingly, the use of artificial intelligence. These requirements are rapidly evolving, may be interpreted and applied inconsistently across jurisdictions, and may conflict with one another, creating uncertainty and increasing the complexity and cost of compliance.
We are also subject to contractual obligations and internal policies governing the handling of personal information. Any actual or perceived failure by us or by our collaborators, service providers or contractors to comply with applicable laws, regulations, contractual obligations or our policies could result in governmental investigations, enforcement actions, litigation, fines, penalties, or other liability, as well as reputational harm, negative publicity and diversion of management time and resources. In addition, such failures could impair our ability to process personal information, conduct clinical trials, operate in certain jurisdictions, or pursue certain business initiatives.
In particular, our information technology systems and those of our contract research organizations, contract manufacturers, collaborators and other third-party service providers are vulnerable to damage or interruption from computer viruses, ransomware and other malicious code, unauthorized access by hackers or sophisticated nation-state and nation-state-supported actors, denial-of-service attacks, employee error or malfeasance, natural disasters and telecommunication and electrical failures. The loss of clinical trial data from completed or ongoing clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. Under SEC rules, we are required to disclose material cybersecurity incidents on Form 8-K within four business days of determining that an incident is material, and any failure to timely identify and disclose such an incident could result in SEC enforcement actions, litigation and reputational damage. We may not have adequate insurance coverage for security
incidents or breaches, and the successful assertion of one or more large claims against us that exceeds our available insurance coverage could have an adverse effect on our business.
Compliance with these requirements is costly and time-consuming and may require us to implement additional safeguards, modify our data practices, or limit our use of data, which could adversely affect our operations. Further, evolving laws governing international data transfers and data localization may restrict our ability to transfer personal information across borders or require us to incur additional costs to do so.
We also face risks related to cybersecurity incidents, including unauthorized access, loss or disclosure of personal or confidential information. Any such incident, whether actual or perceived, could result in legal liability, regulatory scrutiny, operational disruption and reputational damage.
In addition, our increasing use of artificial intelligence and other emerging technologies may subject us to new and developing regulatory requirements and increase risks related to data privacy, cybersecurity, intellectual property and the accuracy and reliability of outputs. Any failure to comply with applicable requirements or to manage these risks could adversely affect our business, financial condition and results of operations.
Significant changes or developments in US laws or policies, including changes in US healthcare regulation, will affect our business and may have a material adverse effect on our business.
There is uncertainty surrounding potential changes to the regulatory environment in the US, particularly as it relates to healthcare regulation and related programs, which may have an adverse effect on our business. For example, the current administration issued an executive order establishing an agency to reform federal government processes and reduce expenditures and has committed to significantly reduce government spending through cuts to federal healthcare programs and reductions in the workforces of key government agencies, such as the US Department of Health & Human Services (HHS), FDA, and CMS. Pressures on and uncertainty surrounding the US federal government’s budget, and potential changes in budgetary priorities, could adversely affect the funding for individual programs, including Medicare and other government programs upon which our business depends. Moreover, further efforts by the current administration to limit federal agency budgets or personnel may lead to slower response times, less guidance and longer review periods, inconsistencies in execution of federal policies, potentially affecting our ability to progress development of our product candidates or obtain regulatory approval for our product candidates. Additionally, in February 2025 HHS ended a longstanding commitment to voluntarily comply with notice-and-comment rulemaking procedures for public benefits rules, even when not required by statute, which could contribute to rapid changes in policy without opportunity for public input. The continuing effects of the US federal government shutdown in late 2025 may delay inspections, reviews or other regulatory activities, and may significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business.
Additionally, further changes in legislation and regulations (including those related to taxation, trade and importation), economic and monetary policies, geopolitical matters, among other potential impacts, could adversely impact the global economy and our operating results. The potential impact of new policies that may be implemented as a result of the current administration is currently uncertain.
The biopharmaceutical industry is subject to extensive regulatory obligations and policies that are subject to significant and abrupt change, including due to judicial challenges, election cycles, and resulting regulatory updates and changes in policy priorities.
On June 28, 2024, the US Supreme Court issued an opinion in Loper Bright Enterprises v. Raimondo holding that courts reviewing agency action pursuant to the Administrative Procedure Act (APA) “must exercise their independent judgment” and “may not defer to an agency interpretation of the law simply because a statute is ambiguous.” The decision has impacted how lower courts evaluate challenges to agency interpretations of law, including those by HHS, CMS, FDA and other agencies with significant oversight of the biopharmaceutical industry. The new framework is likely to increase both the frequency of such challenges and their odds of success by eliminating one way in which the government previously prevailed in such cases. As a result, significant regulatory policies are subject to increased prospects of litigation and judicial scrutiny.
In addition, federal agency activities, priorities, leadership, policies, rulemaking, communications, spending and staffing may be significantly impacted by election cycles and legislative developments. For example, the current US presidential administration has committed to significantly reduce government spending through cuts to federal healthcare programs and reductions in the workforces of key government agencies, such as HHS, FDA, and CMS. Further efforts by the current administration to reduce federal spending may result in reductions to agency budgets, employees, and
operations, which may lead to slower response times, less guidance and longer review periods, potentially affecting our ability to progress development of our product candidates or obtain regulatory approval for our product candidates. The administration and agencies have also made abrupt announcements about new or changed regulatory policies, such as policies related to use of AI to review product applications. These developments may lead to greater uncertainty regarding FDA policies, slower response times, longer review periods, unexpected delays, increased costs, or other negative impacts on our business that are difficult to predict. These changes may potentially affect our ability to progress development of our product candidates or obtain regulatory approval for our product candidates.
Enhanced governmental and public scrutiny over, or investigations or litigation involving, pharmaceutical manufacturer donations to patient assistance programs may require us to modify our programs and could negatively impact our business practices, harm our reputation, divert the attention of management and increase our expenses.
To help patients afford our products, we have a manufacturer-sponsored patient assistance program that helps eligible patients in the US access our therapies. This type of program has become the subject of enforcement scrutiny in recent years. For example, some pharmaceutical manufacturers have been named in lawsuits challenging the legality of their patient assistance programs under a variety of federal and state laws. In addition, certain state and federal enforcement authorities continue to pursue investigations and enter into settlements related to manufacturers’ support of patient assistance programs, and members of Congress have also initiated inquiries on topics that include, for example, manufacturer-sponsored patient assistance programs, co-payment assistance programs, and manufacturer contributions to independent charitable patient assistance programs. Moreover, the HHS, Office of the Inspector General continues to publish advisory opinions and other agency guidance on the topic of patient assistance, which reflects the government’s continued scrutiny of manufacturer sponsored or supported patient assistance programs. Numerous organizations, including pharmaceutical manufacturers, have been subject to ongoing litigation, enforcement activities and settlements related to their patient support programs and certain of these organizations have entered into, or have otherwise agreed to, significant civil settlements with applicable enforcement authorities. It is possible that future legislation may be proposed that would establish requirements or restrictions with respect to these programs and/or support that would affect pharmaceutical manufacturers.
Our patient assistance program could become the target of similar inquiries, litigation, enforcement, and/or legislative proposals. If we are deemed not to have complied with laws or regulations in the operation of, or our interactions with, these programs, we could be subject to damages, fines, penalties or other criminal, civil or administrative sanctions or enforcement actions. We cannot ensure that our compliance controls, policies and procedures will be sufficient to protect against acts of our employees, business partners or vendors that may violate the laws or regulations of the jurisdictions in which we operate. A government investigation could negatively impact our business practices, harm our reputation, divert the attention of management and increase our expenses.
If manufacturers obtain approval for generic versions of our products, or of products with which we compete, our business may be harmed.
Under the Federal Food, Drug and Cosmetic Act (FDCA), the FDA can approve an Abbreviated New Drug Application (ANDA) for a generic version of a branded drug without the ANDA applicant undertaking the clinical testing necessary to obtain approval to market a new drug. Generally, in place of such clinical studies, an ANDA applicant usually needs only to submit data demonstrating that its product has the same active ingredient(s), strength, dosage form and route of administration and that it is bioequivalent to the branded product. In September 2019, the FDA published product-specific bioequivalence guidance on fostamatinib disodium to let potential ANDA applicants understand the data FDA would expect to see for approval of a generic version of our products.
The FDCA requires that an applicant for approval of a generic form of a branded drug certify either that its generic product does not infringe any of the patents listed by the owner of the branded drug in the FDA’s Orange Book or that those patents are not enforceable. This process is known as a paragraph IV challenge. Upon notice of a paragraph IV challenge, a patent owner has 45 days to bring a patent infringement suit in federal district court against the company seeking ANDA approval of a product covered by one of the owner’s patents. If this type of suit is commenced, the FDCA provides a 30-month stay on the FDA’s approval of the competitor’s application. If the litigation is resolved in favor of the ANDA applicant or the challenged patent expires during the 30-month stay period, the stay is lifted, and the FDA may thereafter approve the application based on the standards for approval of ANDAs. Once an ANDA is approved by the FDA, the generic manufacturer may market and sell the generic form of the branded drug in competition with the branded medicine.
The ANDA process can result in generic competition if the patents at issue are not upheld or if the generic competitor is found not to infringe the owner’s patents. If this were to occur with respect to our products or products with which it competes, our business would be harmed.
In March 2025, we entered into a settlement agreement with Annora resolving patent litigation related to our product TAVALISSE. The litigation resulted from submission by Annora of an ANDA to the FDA seeking approval to market a generic version of TAVALISSE in the US. For more information, see “Part I, Item 3, Legal Proceedings” of the Annual Report on Form 10-K for the year ended December 31, 2025.
We intend to vigorously enforce and defend our intellectual property related to our products. We cannot be assured that we will prevent the introduction of a generic version of our products for any particular length of time, or at all. If an ANDA from generic manufacturers is approved, and generic versions of our products are introduced, whether following the expiration of our patents, the invalidation of our patents as a result of any litigation, or the determination that the proposed generic product does not infringe on our patents, our sales of our products would be adversely affected. In addition, we cannot predict what additional ANDAs could be filed by potential generic competitors requesting approval to market generic forms of our products, which would require us to incur significant additional expense and result in distraction for our management team, and if approved, result in significant decreases in the revenue derived from sales of our marketed products and thereby materially harm our business and financial condition.
Unforeseen safety issues could emerge with our products that could require us to change the prescribing information to add warnings, limit use of the product, and/or result in litigation. Any of these events could have a negative impact on our business.
Discovery of unforeseen safety problems or increased focus on a known problem could impact our ability to commercialize our products and could result in restrictions on its permissible uses, including withdrawal of the medicine from the market.
If we or others identify additional undesirable side effects caused by our products after approval:
•regulatory authorities may require the addition of labeling statements, specific warnings, contraindications, Dear Healthcare Provider letters, press releases, field alerts, or other communications containing warnings or other safety information about our products to physicians and pharmacies;
•regulatory authorities may withdraw their approval of the product and require us to take our approved drugs off the market or suspend their commercialization until the identified issues have been satisfactorily addressed;
•we may be required to change the way the product is administered, conduct additional clinical trials, change the labeling of the product, or implement a Risk Evaluation and Mitigation Strategy (REMS);
•we may have additional limitations on how we promote our drugs;
•third-party payors may limit coverage or reimbursement for our products;
•sales of our products may decrease significantly;
•we may be subject to litigation or product liability claims; and
•our reputation may suffer.
Any of these events could prevent us from achieving or maintaining market acceptance of our products and could substantially increase our operating costs and expenses, which in turn could delay or prevent us from generating significant revenue from sale of our products. For example, in October 2024, we issued a Dear Healthcare Provider letter related to a new safety signal for GAVRETO. The letter advises healthcare providers to apply certain measures to protect patient safety, including enhanced ongoing monitoring for signs and symptoms of infection as well as guidance for withholding treatment to patients in the presence of active infection. On December 22, 2025, the FDA notified us of the approval of a Prior Approval supplemental NDA for GAVRETO, which updated the US Prescribing Information to add a boxed warning regarding serious infections, including opportunistic infections. The addition of a boxed warning is the most prominent safety warning the FDA can require and could negatively affect prescribing rates, patient willingness to initiate or continue
therapy, and payer coverage and reimbursement for GAVRETO. If the box warning causes healthcare providers to prescribe GAVRETO less frequently, or if payers impose additional restrictions on coverage results, the commercial success of GAVRETO or any of our other drug products could be limited.
Side effects and toxicities associated with our products, as well as the warnings, precautions and requirements listed in the prescribing information for our products, could affect the willingness of physicians to prescribe, and patients to utilize, our products and thus harm commercial sales of our products. For example, for REZLIDHIA, the FDA-approved label contains a boxed warning describing the risk of differentiation syndrome, which can be fatal, in patients receiving the drug. This and other restrictions could limit the commercial success of the product.
If a safety issue emerges post-approval, we may become subject to costly product liability litigation by our customers, their patients or payors. Product liability claims could divert management’s attention from our core business, be expensive to defend, and result in sizable damage awards against us that may not be covered by insurance. If we cannot successfully defend ourselves against claims that our products caused injuries, we will incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
•decreased demand for any product candidates or products that we may develop;
•the inability to commercialize any products that we may develop;
•injury to our reputation and significant negative media attention;
•withdrawal of patients from clinical studies or cancellation of studies;
•significant costs to defend the related litigation;
•substantial monetary awards to patients; and
•loss of revenue.
We currently hold $10.0 million in product liability insurance coverage, which may not be adequate to cover all liabilities that we may incur. Insurance coverage is increasingly expensive. We may not be able to obtain insurance coverage at a reasonable cost or in amounts adequate to satisfy any liability or associated costs that may arise in the future. These events could harm our business and results of operations and cause our stock price to decline.
Our business could be materially and adversely affected by pandemics or other public health crises.*
Pandemics or similar public health events, and governmental responses thereto, may disrupt our commercialization efforts, clinical development activities, supply chain, regulatory interactions and overall operations. These disruptions may reduce demand for our products, delay or impair clinical trials, interrupt the manufacture and supply of our products and clinical materials, and affect the ability of our collaborators and service providers to perform their obligations.
Pandemics may also delay regulatory review, inspections and approvals, and could negatively impact global economic conditions and our access to capital. The extent of any such impacts is uncertain and beyond our control. Any of these effects could materially and adversely affect our business, financial condition, results of operations and prospects, and may exacerbate other risks described in this section.
If we fail to comply with our reporting and payment obligations under the Medicaid Drug Rebate Program or other governmental pricing programs in the US, we could be subject to additional rebate or discount requirements, fines, sanctions and exposure under other laws which could have an adverse effect on our business, results of operations and financial condition.
We participate in the Medicaid Drug Rebate Program, as administered by CMS, the 340B Drug Pricing Program, as administered by the Health Resources and Services Administration (HRSA), and other federal and state government drug pricing programs in the US, and we may participate in additional government pricing programs in the future. These programs generally require us to pay rebates or otherwise provide discounts to government payors and/or required covered entities in connection with drugs that are dispensed to beneficiaries/recipients of these programs. In some cases, such as
with the Medicaid Drug Rebate Program, the rebates are based on pricing metrics that we report on a monthly and quarterly basis to the government agencies that administer the programs. Pricing requirements and rebate/discount calculations are complex, vary among products and programs, and are often subject to interpretation by governmental or regulatory agencies and the courts. The requirements of these programs, including, by way of example, their respective terms and scope, change frequently. Responding to current and future changes may increase our costs, and ensuring compliance will be time consuming. Invoicing for rebates is provided in arrears, and there is frequently a time lag of up to several months between the sales to which rebate notices relate and our receipt of those notices, which further complicates our ability to accurately estimate and accrue for rebates related to the Medicaid program as implemented by individual states. Thus, there can be no assurance that we will be able to identify all factors that may cause our discount and rebate payment obligations to vary from period to period, and our actual results may differ significantly from our estimated allowances for discounts and rebates. Changes in estimates and assumptions may have an adverse effect on our business, results of operations and financial condition.
In addition, the HHS, Office of Inspector General and other governmental enforcement and administrative bodies have increased their focus, including through recent enforcement actions against manufacturers, on pricing requirements for products, including, but not limited to the methodologies used by manufacturers to calculate average manufacturer price and best price for compliance with reporting requirements under the Medicaid Drug Rebate Program. We are liable for errors associated with our submission of pricing data and for any overcharging of government payors. Failure to make necessary disclosures and/or to identify overpayments could result in allegations against us under the federal False Claims Act and other laws and regulations. Any required refunds to the US government or response to a government investigation or enforcement action would be expensive and time consuming and could have an adverse effect on our business, results of operations and financial condition. In addition, in the event that CMS were to terminate our rebate agreement, no federal payments would be available under Medicaid for our covered outpatient drugs or under Medicare Part B for any of our products that may be reimbursed under Part B.
Finally, we may be affected by developments relating to the 340B Drug Pricing Program (340B Program). Multiple states have recently enacted or are currently considering laws that require manufacturers to ship 340B drugs to certain contract pharmacies and impose various civil and criminal penalties on manufacturers that do not comply. These laws have been challenged in federal court and many of the cases are pending. In March 2024, the US Court of Appeals for the Eight Circuit upheld the Arkansas law prohibiting drug makers for restricting 340B drug discounts for providers using contract pharmacies. HHS also issued a final rule on procedures for the 340B Program’s administrative dispute resolution process in April 2024. Additionally, under the Trump administration, several changes to the 340B program have been considered, including a proposal in the President’s 2026 budget to shift oversight of the 340B program from the HRSA to CMS. Additionally, on July 31, 2025, the HRSA announced that it will implement a 340B Rebate Model Pilot Program that will be open to a selected group of drugs and manufacturers. However, the HRSA withdrew this proposal in January 2026 following litigation that resulted in a federal court granting temporary restraining order to block the program and has since requested information from stakeholders as to alternative rebate models. It is unclear how the other pending litigation, proposed legislation, or future administrative action relating to the 340B Program will impact our business.
Even for those product candidates that have or may receive regulatory approval, they may fail to achieve the degree of market acceptance by physicians, patients, third-party payors and others in the medical community necessary for commercial success, in which case we may not generate significant revenues or become profitable.
For our product candidates that have or may receive regulatory approval, they may nonetheless fail to gain sufficient market acceptance by physicians, hospital administrators, patients, third-party payors and others in the medical community. The degree of market acceptance of our product candidates, if approved for commercial sale, will depend on a number of factors, including the following:
•relative convenience and ease of administration;
•the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies;
•the willingness of physicians to change their current treatment practices;
•any additional support that may be required to administer the treatment to patients;
•the willingness of hospitals and hospital systems to include our product candidates as treatment options;
•demonstration of efficacy and safety in clinical trials;
•the prevalence and severity of any side effects;
•the ability to offer product candidates for sale at competitive prices;
•the price we charge for our product candidates;
•the strength of marketing and distribution support; and
•the availability of third-party coverage and adequate reimbursement and the willingness of patients to pay out-of-pocket in the absence of such coverage and adequate reimbursement.
Efforts to educate the physicians, patients, third-party payors and others in the medical community on the benefits of our product candidates may require significant resources and may not be successful. If any of our product candidates are approved, but do not achieve an adequate level of acceptance, we may not generate significant product revenue and we may not become profitable on a sustained basis.
We may need to seek additional capital in the future to sufficiently fund our operations, research and corporate development opportunities.
We have consumed substantial amounts of capital to date as we continue our research and development activities, including preclinical studies and clinical trials and for the commercialization of our products, as well as corporate development opportunities. We may seek another collaborator or licensee in the future for further clinical development and commercialization of our products, as well as our other clinical programs, which we may not be able to obtain on commercially reasonable terms or at all. We believe that our existing capital resources will be sufficient to support our current and projected funding requirements, including the continued commercialization of our products through at least the next 12 months. We have based this estimate on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we currently expect. Because of the numerous risks and uncertainties associated with commercial launch, the development of our product candidates and other research and development activities, we are unable to estimate with certainty our future product revenues, our revenues from our current and future collaborative partners, the amounts of increased capital outlays and operating expenditures associated with our current and anticipated clinical trials and other research and development activities.
While we intend to opportunistically seek access to additional funds through public or private equity offerings or debt financings, we do not know whether additional financing will be available when needed, or that, if available, we will obtain financing on reasonable terms. Our ability to raise additional capital, including our ability to secure new collaborations and continue to support existing collaboration efforts with our partners, may also be adversely impacted by potential worsening global economic conditions and the recent disruptions to, and volatility in, the credit and financial markets in the US and worldwide resulting from global geopolitical tensions. Unless and until we are able to generate a sufficient amount of product, royalty or milestone revenue, which may never occur, we expect to finance future cash needs through public and/or private offerings of equity securities, debt financings or collaboration and licensing arrangements, as well as through proceeds from the exercise of stock options and interest income earned on the investment of our cash balances and short-term investments. To the extent we raise additional capital by issuing equity securities in the future, our stockholders could at that time experience substantial dilution. In addition, we have a significant number of stock options outstanding. To the extent that outstanding stock options have been or may be exercised or other shares issued, our stockholders may experience further dilution. Further, we may choose to raise additional capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. Our credit facility with MidCap includes certain covenants that may restrict our business, and any other debt financing that we are able to obtain in the future may involve operating covenants that restrict our business. To the extent that we raise additional funds through any new collaboration and licensing arrangements, we may be required to refund certain payments made to us, relinquish some rights to our technologies or product candidates or grant licenses on terms that are not favorable to us.
We have indebtedness in the form of a term loan pursuant to the Credit Agreement with MidCap, which could adversely affect our financial condition and our ability to respond to changes in our business. Further, if we are unable to satisfy certain conditions of the Credit Agreement, we will be unable to draw down the remainder of the facility.*
We entered into a Credit Agreement with MidCap on September 27, 2019, amended on March 29, 2021, February 11, 2022, July 27, 2022, and April 11, 2024. The Credit Agreement provides for a $60.0 million term loan credit facility. As of March 31, 2026, the outstanding principal balance of the loan was $45.0 million. Under the Credit Agreement, we
were required to repay amounts due when there is an event of default for the term loans that results in the principal, premium, if any, and interest, if any, becoming due prior to the maturity date for the term loans. The Credit Agreement also contains a number of other affirmative and restrictive covenants. These and other terms had to be monitored closely for compliance and could have restricted our ability to grow our business or enter into transactions that we believe would be beneficial to our business.
On May 5, 2026, we terminated the term loan facility and entered into a new Credit Agreement with MidCap providing for a revolving credit facility with a maximum borrowing capacity of $40.0 million, with an option to increase to $60.0 million, subject to customary conditions. As of the date of this filing, we had an outstanding borrowing of $8.0 million under the revolving credit facility. Borrowings under the revolving credit facility are subject to availability and ongoing compliance with certain conditions, and we may not be able to access the full amount of the facility when needed. Availability under the revolving credit facility is subject to a borrowing base based primarily on eligible accounts receivable and inventory. While the revolving credit facility enhances our financial flexibility to support operations and working capital needs, our liquidity is dependent on the level of borrowing base availability. In addition, the revolving credit facility may require us to seek additional financing over time to support our operations, and such financing may not be available on favorable terms, or at all.
The revolving credit facility bears interest at a variable rate based on SOFR, subject to a floor, which exposes us to the risk of increased interest expense in a rising interest rate environment. The Credit Agreement contains affirmative and restrictive covenants, including financial covenants. These and other terms must be monitored closely for compliance and could restrict our ability to grow our business or enter into transactions that we believe would be beneficial to our business.
Our business may not generate cash flow from operations in the future sufficient to service our debt and support our growth strategies. If we are unable to generate such cash flow, we may be required to adopt one or more alternatives, such as restructuring our debt or obtaining additional equity capital on terms that may be onerous or highly dilutive. Our ability to refinance our indebtedness will depend on the capital markets and our financial condition at such time. We may not be able to engage in any of these activities or engage in these activities on desirable terms, which could result in a default on our current debt obligations. In addition, we cannot be sure that additional financing will be available when required or, if available, will be on terms satisfactory to us. Further, even if we are able to obtain additional financing, we may be required to use such proceeds to repay amounts outstanding under our Credit Agreement.
Our indebtedness may have other adverse effects, such as:
•our vulnerability to adverse general economic conditions and heightened competitive pressures;
•dedication of a portion of our cash flow from operations to interest payments, limiting the availability of cash for other operational purposes;
•limited flexibility in planning for, or reacting to, changes in our business and industry; and
•our inability to obtain additional financing in the future.
The Credit Agreement contains provisions that could result in an event of default, including a mandatory prepayment provision that gives MidCap and/or its agent the right to demand payment of any outstanding borrowings, together with applicable interest and fees, upon the occurrence of an event of default. If we fail to comply with the covenants or other requirements under the revolving credit facility, an event of default could occur, which could result in the acceleration of any outstanding borrowings and the exercise of remedies by the lender, including foreclosure on substantially all of our assets. We may not have sufficient available cash or be able to obtain financing at the time we are required to repay amounts outstanding under the Credit Agreement.
We rely and may continue to rely on two distribution facilities for the sale of our products and potential sale of any of our product candidates.
Our distribution operations for the sale of our products are currently concentrated in two distribution centers owned by a third-party logistics provider. Additionally, our distribution operations, if and when we launch any of our product candidates in the future, may also be concentrated in such distribution centers owned by a third-party logistics provider. Any errors in inventory level management and unforeseen inventory shortage could adversely affect our business.
In addition, any significant disruption in the operation of the facility due to natural disaster or severe weather, or events such as fire, accidents, power outages, system failures, or other unforeseen causes, could devalue or damage a significant portion of our inventories and could adversely affect our product distribution and sales until such time as we could secure an alternative facility. Further, climate change may increase both the frequency and severity of extreme weather conditions and natural disasters, which may affect our business operations. If we encounter difficulties with any of our distribution facilities, whether due to the potential future impacts of a global pandemic (including as a result of disruptions of global shipping and the transport of products) or otherwise, or other problems or disasters arise, we cannot ensure that critical systems and operations will be restored in a timely manner or at all, and this would have an adverse effect on our business. In addition, growth could require us to further expand our current facility, which could affect us adversely in ways that we cannot predict.
Forecasting potential sales for any of our product candidates will be difficult, and if our projections are inaccurate, our business may be harmed, and our stock price may be adversely affected.
Our business planning requires us to forecast or make assumptions regarding product demand and revenues for any of our product candidates if they are approved despite numerous uncertainties. These uncertainties may be increased if we rely on our collaborators or other third parties to conduct commercial activities in certain geographies and provide us with accurate and timely information. Actual results may differ materially from projected results for various reasons, including the following, as well as risks identified in other risk factors:
•the efficacy and safety of any of our product candidates, including as relative to marketed products and product candidates in development by third parties;
•pricing (including discounting or other promotions), reimbursement, product returns or recalls, competition, labeling, adverse events and other items that impact commercialization;
•the rate of adoption in the particular market, including fluctuations in demand for various reasons;
•potential future impacts, if any, including a global pandemic;
•lack of patient and physician familiarity with the drug;
•lack of patient use and physician prescribing history;
•lack of commercialization experience with the drug;
•actual sales to patients may significantly differ from expectations based on sales to wholesalers; and
•uncertainty relating to when the drug may become commercially available to patients and rate of adoption in other territories.
We expect that our revenues from sales of any of our products will continue to be based in part on estimates, judgment and accounting policies. Any incorrect estimates or disagreements with regulators or others regarding such estimates or accounting policies may result in changes to our guidance, projections or previously reported results. We make estimates for provisions for sales discounts, returns and allowances. Our estimates are based on available customer and payor data received from the specialty pharmacies and distributors, as well as third party market research data. In part, our estimates are dependent on our distribution channel and payor mix. If actual results in the future vary from our estimates, we adjust these estimates, which would affect our net product revenue and earnings in the period such variances become known. Expected and actual product sales and quarterly and other results may greatly fluctuate, including in the near-term, and such fluctuations can adversely affect the price of our common stock, perceptions of our ability to forecast demand and revenues, and our ability to maintain and fund our operations.
We do not and will not have access to all information regarding our products and product candidates we licensed to our collaboration partners.
We do not and will not have access to all information regarding our products and other product candidates, including potentially material information about commercialization plans, medical information strategies, clinical trial design and execution, safety reports from clinical trials, safety reports, regulatory affairs, process development,
manufacturing and other areas known by our collaboration partners. In addition, we have confidentiality obligations under our respective agreements with our collaboration partners. Thus, our ability to keep our shareholders informed about the status of our products and other product candidates will be limited by the degree to which our collaboration partners keep us informed and allows us to disclose such information to the public. If our collaboration partners fail to keep us informed about commercialization efforts related to our products, or the status of the clinical development or regulatory approval pathway of other product candidates licensed to them, we may make operational and/or investment decisions that we would not have made had we been fully informed, which may adversely affect our business and operations.
Our future funding requirements will depend on many uncertain factors.
Our future funding requirements will depend upon many factors, many of which are beyond our control, including, but not limited to:
•the costs to commercialize our products in the US, or any other future product candidates, if any such candidate receives regulatory approval for commercial sale;
•the progress and success of our clinical trials and preclinical activities (including studies and manufacture of materials) of our product candidates conducted by us;
•our ability to secure patent and regulatory protection;
•our ability to secure a favorable price or a positive HTA assessment;
•potential future impacts, if any, of a global pandemic;
•the costs and timing of regulatory filings and approvals by us and our collaborators;
•the progress of research and development programs carried out by us and our collaborative partners;
•any changes in the breadth of our research and development programs;
•the ability to achieve the events identified in our collaborative agreements that may trigger payments to us from our collaboration partners;
•our ability to acquire or license other technologies or compounds that we may seek to pursue;
•our ability to manage our growth;
•competing technological and market developments;
•the costs and timing of obtaining, enforcing and defending our patent and other intellectual property rights; and
•expenses associated with any unforeseen litigation, including any arbitration and securities class action lawsuits.
Insufficient funds may require us to delay, scale back or eliminate some or all of our commercial efforts and/or research and development programs, to reduce personnel and operating expenses, to lose rights under existing licenses or to relinquish greater or all rights to product candidates at an earlier stage of development or on less favorable terms than we would otherwise choose or may adversely affect our ability to operate as a going concern.
Our recent operating income may not be sustainable, and we may continue to incur significant losses.
For the three months ended March 31, 2026, in 2025 and 2024, we recognized income from operations primarily due to net product sales and collaboration revenues, partially offset by our operating expenses. Historically, we have incurred losses from operations each year since we were incorporated in June 1996 other than in fiscal year 2010, due in large part to the significant research and development expenditures and costs of our ongoing commercial efforts. Although we are now recognizing income from operations, there can be no assurance that we will continue to generate annual
operating income in the foreseeable future. Currently, our potential sources of revenues include sales of our products, as well as upfront, milestones and royalty payments pursuant to our collaboration arrangements, all of which may never materialize if sales of our products decline or if our collaboration partners do not achieve certain events or generate net sales to which these contingent payments are dependent on. If our future drug candidates fail or do not gain regulatory approval, or if our drugs do not achieve sustainable market acceptance, we may not be profitable. As of March 31, 2026, we had an accumulated deficit of approximately $1.0 billion. The extent of our future losses or profitability, if any, is uncertain.
If our corporate collaborations or license agreements are unsuccessful, or if we fail to form new corporate collaborations or license agreements, our research and development efforts could be delayed.
Our strategy depends upon the formation and sustainability of multiple collaborative arrangements and license agreements with third parties now and in the future. We rely on these arrangements for not only financial resources, but also for expertise we need now and in the future relating to clinical trials, manufacturing, sales and marketing, and for licenses to technology rights. To date, we have entered into several such arrangements with corporate collaborators; however, we do not know if these collaborations or additional collaborations with third parties, if any, will dedicate sufficient resources or if any development or commercialization efforts by third parties will be successful. In addition, our corporate collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a drug candidate or development program. Should a collaborative partner fail to develop or commercialize a compound or product to which it has rights from us for any reason, including corporate restructuring, such failure might delay our ongoing research and development efforts, because we might not receive any future payments, and we would not receive any royalties associated with such compound or product. We may seek another collaborator or licensee in the future for clinical development and commercialization of our products, as well as our other clinical programs, which we may not be able to obtain on commercially reasonable terms or at all. If we are unable to form new collaborations or enter into new license agreements, our research and development efforts could be delayed. In addition, the continuation of some of our partnered drug discovery and development programs may be dependent on the periodic renewal of our corporate collaborations.
Each of our collaborations could be terminated by the other party at any time, and we may not be able to renew these collaborations on acceptable terms, if at all, or negotiate additional corporate collaborations on acceptable terms, if at all. If these collaborations terminate or are not renewed, any resultant loss of revenues from these collaborations or loss of the resources and expertise of our collaborative partners could adversely affect our business. For example, in April 2026, we received written notice from Lilly of its election to terminate the Lilly Agreement, which will become effective June 15, 2026. Following termination of the Lilly Agreement, including the prior termination of the CNS disease program effective in November 2025, we do not expect to receive future milestones or royalties under the Lilly Agreement.
Conflicts also might arise with collaborative partners concerning proprietary rights to particular compounds. While our existing collaborative agreements typically provide that we retain milestone payments, royalty rights and/or revenue sharing with respect to drugs developed from certain compounds or derivative compounds, any such payments or royalty rights may be at reduced rates, and disputes may arise over the application of payment provisions or derivative payment provisions to such drugs, and we may not be successful in such disputes. Additionally, the management teams of our collaborators may change for various reasons including due to being acquired. Different management teams or an acquiring company of our collaborators may have different priorities which may have adverse results on the collaboration with us.
We are also a party to various license agreements that give us rights to use specified technologies in our research and development processes. The agreements pursuant to which we have in-licensed technology permit our licensors to terminate the agreements under certain circumstances. If we are not able to continue to license these and future technologies on commercially reasonable terms, our product development and research may be delayed or otherwise adversely affected.
If conflicts arise between our collaborators or advisors and us, any of them may act in their self-interest, which may be adverse to our stockholders’ interests.
If conflicts arise between us and our corporate collaborators or scientific advisors, the other party may act in its self-interest and not in the interest of our stockholders. Some of our corporate collaborators are conducting multiple product development efforts within each disease area that is the subject of the collaboration with us or may be acquired or merged with a company having a competing program. In some of our collaborations, we have agreed not to conduct, independently or with any third party, any research that is competitive with the research conducted under our
collaborations. Our collaborators, however, may develop, either alone or with others, products in related fields that are competitive with the products or potential products that are the subject of these collaborations. Competing products, either developed by our collaborators or to which our collaborators have rights, may result in their withdrawal of support for our product candidates.
If any of our corporate collaborators were to breach or terminate its agreement with us or otherwise fail to conduct the collaborative activities successfully and in a timely manner, the preclinical or clinical development or commercialization of the affected product candidates or research programs could be delayed or terminated. We generally do not control the amount and timing of resources that our corporate collaborators devote to our programs or potential products. We do not know whether current or future collaborative partners, if any, might pursue alternative technologies or develop alternative products either on their own or in collaboration with others, including our competitors, as a means for developing treatments for the diseases targeted by collaborative arrangements with us.
Our success is dependent on intellectual property rights held by us and third parties, and our interest in such rights is complex and uncertain.
Our success will depend to a large part on our own, our licensees’ and our licensors’ ability to obtain and defend patents for each party’s respective technologies and the compounds and other products, if any, resulting from the application of such technologies. For example, fostamatinib is covered as a composition of matter in a US issued patent that has an expiration date of September 2031, olutasidenib is covered as a composition of matter in a US issued patent that has an expected expiration date of December 2036, after taking into account patent term extension rules, and pralsetinib is covered as a composition of matter in a US issued patent that has an expiration date in November 2036 and subject to extensions.
In the future, our patent position might be highly uncertain and involve complex legal and factual questions, and the cost to defend may also be significant. For example, we may be involved in post-grant proceedings before the US Patent and Trademark Office. Post-grant proceedings are complex and expensive legal proceedings and there is no assurance we will be successful in any such proceedings. A post-grant proceeding could result in our losing our patent rights and/or our freedom to operate and/or require us to pay significant royalties. Additionally, third parties may challenge the validity, enforceability or scope of our issued patents, which may result in such patents being narrowed, invalidated or held unenforceable through interference, opposition or invalidity proceedings before the US Patent and Trademark Office or non-US patent offices. Any successful opposition to our patents could deprive us of exclusive rights necessary for the successful commercialization of our products or our other product candidates. Oppositions could also be filed to complementary patents, such as formulations, methods of manufacture and methods of use, that are intended to extend the patent life of the overall portfolio beyond the patent life covering the composition of matter. A successful opposition to any such complementary patent could impact our ability to extend the life of the overall portfolio beyond that of the related composition of matter patent.
An adverse outcome may allow third parties to use our intellectual property without a license and/or allow third parties to introduce generic and other competing products, any of which would negatively impact our business. For example, in March 2025, we entered into a settlement agreement with Annora resolving patent litigation related to our product TAVALISSE. For more information, see “Part I, Item 3, Legal Proceedings” of our Annual Report on Form 10-K for the year ended December 31, 2025.
We intend to vigorously enforce and defend our intellectual property rights related to our products. Should any third parties receive FDA approval of an ANDA for a generic version of our products or a 505(b)(2) NDA with respect to our products, and if our patents covering our products were held to be invalid (or if such competing generic versions of our products were found to not infringe our patents), then they could introduce generic versions of our products or other such 505(b)(2) products before our patents expire, and the resulting competition would negatively affect our business, financial condition and results of operations. Please also see the risk factor entitled, “If manufacturers obtain approval for generic versions of our products, or of products with which we compete, our business may be harmed.” In the future, there might be other claims that are subject to substantial uncertainties and unascertainable damages or other remedies, and the cost to defend may also be significant.
Additional uncertainty may result because no consistent policy regarding the breadth of legal claims allowed in biotechnology patents has emerged to date. Accordingly, we cannot predict the breadth of claims allowed in our or other companies’ patents.
Because the degree of future protection for our proprietary rights is uncertain, we cannot assure that:
•we were the first to make the inventions covered by each of our pending patent applications;
•we were the first to file patent applications for these inventions;
•others will not independently develop similar or alternative technologies or duplicate any of our technologies;
•any of our pending patent applications will result in issued patents;
•any patents issued to us or our collaborators will provide a basis for commercially viable products or will provide us with any competitive advantages or will not be challenged by third parties;
•we will develop additional proprietary technologies that are patentable;
•we will obtain a supplementary protection certificate that will extend the protection afforded by the patent to the product with a marketing authorization; or
•the patents of others will not have a negative effect on our ability to do business.
We rely on trade secrets to protect technology where we believe patent protection is not appropriate or obtainable; however, trade secrets are difficult to protect. While we require employees, collaborators and consultants to enter into confidentiality agreements, we may not be able to adequately protect our trade secrets or other proprietary information in the event of any unauthorized use or disclosure or the lawful development by others of such information.
We are a party to certain in-license agreements that are important to our business, and we generally do not control the prosecution of in-licensed technology. Accordingly, we are unable to exercise the same degree of control over this intellectual property as we exercise over our internally developed technology. Moreover, some of our academic institution licensors, research collaborators and scientific advisors have rights to publish data and information in which we have rights. If we cannot maintain the confidentiality of our technology and other confidential information in connection with our collaborations, our ability to receive patent protection or protect our proprietary information may otherwise be impaired. In addition, some of the technology we have licensed relies on patented inventions developed using US government resources.
The US government retains certain rights, as defined by law, in such patents, and may choose to exercise such rights. Certain of our in-licenses may be terminated if we fail to meet specified obligations. If we fail to meet such obligations and any of our licensors exercise their termination rights, we could lose our rights under those agreements. If we lose any of our rights, it may adversely affect the way we conduct our business. In addition, because certain of our licenses are sublicenses, the actions of our licensors may affect our rights under those licenses.
If a dispute arises regarding the infringement or misappropriation of the proprietary rights of others, such dispute could be costly and result in delays in our research and development activities, partnering and commercialization activities.
Our success will depend, in part, on our ability to operate without infringing or misappropriating the proprietary rights of others. There are many issued patents and patent applications filed by third parties relating to products or processes that are similar or identical to our licensors or ours, and others may be filed in the future. There may also be copyrights or trademarks that third parties hold. There can be no assurance that our activities, or those of our licensors, will not violate intellectual property rights of others. We believe that there may be significant litigation in the industry regarding patent and other intellectual property rights, and we do not know if our collaborators or we would be successful in any such litigation. Any legal action against our collaborators or us claiming damages or seeking to enjoin commercial activities relating to the affected products, our methods or processes could:
•require our collaborators or us to obtain a license to continue to use, manufacture or market the affected products, methods or processes, which may not be available on commercially reasonable terms, if at all;
•prevent us from using the subject matter claimed in the patents held by others;
•subject us to potential liability for damages;
•consume a substantial portion of our managerial and financial resources; and
•result in litigation or administrative proceedings that may be costly, whether we win or lose.
Our effective tax rate may fluctuate, and we may incur obligations in tax jurisdictions in excess of accrued amounts.
We are subject to taxation in numerous US states and territories. As a result, our effective tax rate is derived from a combination of applicable tax rates in the various places that we operate. In preparing our financial statements, we estimate the amount of tax that will become payable in each of such jurisdictions. Nevertheless, our effective tax rate may be different than experienced in the past due to numerous factors, including passage of the newly enacted federal income tax law, changes in the mix of our profitability from state to state, the results of examinations and audits of our tax filings, our inability to secure or sustain acceptable agreements with tax authorities, changes in accounting for income taxes and changes in tax laws. Any of these factors could cause us to experience an effective tax rate significantly different from previous periods or our current expectations and may result in tax obligations in excess of amounts accrued in our financial statements.
In July 2025, the One Big Beautiful Bill Act (OBBBA) was signed into law. The OBBBA includes a broad range of provisions affecting business entities, including the establishment of certain permanent business tax measures. Among other changes, the legislation permits permanent and immediate deduction for domestic research and development expenditures and restoration of favorable tax treatment for certain business provisions. The legislation contains multiple effective dates, with certain provisions effective beginning in 2025 and others phased through 2027. In accordance with ASC 740, Income Taxes, the effects of changes in tax laws are recognized in the period of enactment. Accordingly, we evaluated the provisions of the OBBBA and determined that the most significant impact to us relates to the capitalization requirements for research and experimental expenditures under Section 174 of the Internal Revenue Code (Section 174). The effects of this provision have been reflected in our income tax provision in 2025. The effects of the OBBBA on our financial statements were not material, other than the impact related to Section 174 as described above. While we continue to evaluate the impact of these legislative changes as additional guidance becomes available, uncertainty remains regarding the timing and interpretation by tax authorities in affected jurisdictions. These legislative changes could have an adverse impact on our future effective tax rate, tax liabilities, and cash tax.
Our ability to use net operating losses (NOLs) and certain other tax attributes is uncertain and may be limited.
Our ability to use our federal and state NOLs to offset potential future taxable income and related income taxes that would otherwise be due is dependent upon our generation of future taxable income before the expiration dates of the NOLs, and we cannot predict with certainty when, or whether, we will generate sufficient taxable income to use all of our NOLs. Federal NOLs generated prior to 2018 will continue to be governed by the NOL carryforward rules as they existed prior to the adoption of the Tax Cuts and Jobs Act (TCJA), which means that generally they will expire 20 years after they were generated if not used prior thereto. Many states have similar laws. Accordingly, our federal and state NOLs could expire unused and be unavailable to offset future income tax liabilities. Moreover, federal NOLs generated in tax years ending after December 31, 2017 may be carried forward indefinitely, but the deductibility of such federal NOLs may be limited to 80% of current year taxable income for tax years beginning after January 1, 2021. In June 2024, California Senate Bill 167 was signed into law which suspends NOL deductions for tax years beginning on or after January 1, 2024 and before January 1, 2027 for taxpayers with net business income or modified adjusted gross income of at least $1.0 million for the tax year. The legislation also limits the aggregate use of otherwise allowable business credits to $5.0 million for each tax year beginning on or after January 1, 2024 but before January 1, 2027 (except for certain credits not subject to the limitation). Although the TCJA required taxpayers to capitalize research and experimental expenditures under Section 174 for tax years beginning after December 31, 2022, the OBBBA restored and made permanent the ability for taxpayers to make immediate deductions for research and experimental expenditures generated in tax years beginning after December 31, 2024.
In addition, utilization of NOLs to offset potential future taxable income and related income taxes that would otherwise be due is subject to annual limitations under the “ownership change” provisions of Sections 382 and 383 of the Section 174 and similar state provisions, which may result in the expiration of NOLs before future utilization. In general, under Section 174, if a corporation undergoes an “ownership change,” generally defined as a greater than 50% change (by value) in its equity ownership over a three-year period, the corporation’s ability to use its pre-change NOLs and other pre-change tax attributes (such as research and development credit carryforwards) to offset its post-change taxable income or taxes may be limited. Our equity offerings and other changes in our stock ownership, some of which are outside of our control, may have resulted or could in the future result in an ownership change. Although we have completed studies to provide reasonable assurance that an ownership change limitation would not apply, we cannot be certain that a taxing authority would reach the same conclusion. If, after a review or audit, an ownership change limitation were to apply, utilization of our domestic NOLs and tax credit carryforwards could be limited in future periods and a portion of the
carryforwards could expire before being available to reduce future income tax liabilities. Moreover, our ability to utilize our NOLs is conditioned upon us achieving profitability and generating US federal taxable income.
Changes in valuation allowance of deferred tax assets may affect our future operating results
Our deferred tax assets are primarily from net operating loss carryforwards, tax credits and other deductible temporary differences. Historically, we maintained a full valuation allowance on our outstanding deferred tax assets. In the fourth quarter of 2025, based on our evaluation of all available positive and negative evidence, we concluded that it was more-likely-than not that a significant portion of our federal and state deferred tax assets would be realized. Accordingly, we released the valuation allowance against these deferred tax assets, except for deferred tax assets associated with the portion of federal research and development credit carryforwards, California NOL and California research and development credit carryforwards. The assessment of the realizability of deferred tax assets involved considerable management judgment and required evaluation of all available evidence, including cumulative recent financial performance, forecasts of future taxable income, and the reversal of taxable temporary differences. As a result of this assessment, we recognized a deferred income tax benefit of $245.9 million in 2025.
Our evaluation process for deferred tax asset realizability incorporates multiple factors, including, historical earnings performance and trends, projections of future taxable income and associated risks, and strategic business developments and market conditions. We conduct periodic reviews of our deferred tax asset balances to assess continued realizability. When evidence suggests it is more-likely-than-not that deferred tax assets will not be realized, we maintain or establish appropriate valuation allowances. The ultimate realization of our deferred tax assets depends principally on generating sufficient future taxable income during periods when temporary differences reverse. Changes in our assessment can result in material adjustments to the valuation allowance, which directly affects our income tax provision and effective tax rate. The release of a valuation allowance results in a tax benefit, while establishing additional allowances increases tax expense. Changes in projected future performance or shifts in the weighting of positive versus negative evidence can lead to significant changes in the required valuation allowance, making this estimate particularly sensitive to management's judgments about future operations.
Because we expect to be dependent upon collaborative and license agreements, we might not meet our strategic objectives.
Our ability to generate revenue in the near term depends on the timing of recognition of certain upfront payments, achievement of certain payment triggering events with our existing collaboration agreements and our ability to enter into additional collaborative agreements with third parties. Our ability to enter into new collaborations and the revenue, if any, that may be recognized under these collaborations is highly uncertain. If we are unable to enter into one or more new collaborations, our business prospects could be harmed, which could have an immediate adverse effect on our ability to continue to develop our compounds and on the trading price of our stock. Our ability to enter into a collaboration may be dependent on many factors, such as the results of our clinical trials, competitive factors and the fit of one of our programs with another company’s risk tolerance, including toward regulatory issues, patent portfolio, clinical pipeline, the stage of the available data, particularly if it is early, overall corporate goals and financial position.
To date, a portion of our revenues have been related to the research or transition phase of each of our collaborative agreements. Such revenues are for specified periods, and the impact of such revenues on our results of operations is at least partially offset by corresponding research costs. Following the completion of the research or transition phase of each collaborative agreement, additional revenues may come only from payments triggered by milestones and/or the achievement of other contingent events, and royalties, which may not be paid, if at all, until certain conditions are met. This risk is heightened due to the fact that unsuccessful research efforts may preclude us from receiving any contingent payments under these agreements. Our receipt of revenues from collaborative arrangements is also significantly affected by the timing of efforts expended by us and our collaborators and the timing of lead compound identification. We have received payments from our collaborations including Lilly, Grifols, Kissei, Medison, Knight, Dr. Reddy's, BerGenBio, and Daiichi. Under several agreements, future payments may not be earned until the collaborator has advanced product candidates into clinical testing, which may never occur or may not occur until sometime well into the future. If we are not able to generate revenue under our collaborations when and in accordance with our expectations or the expectations of industry analysts, this failure could harm our business and have an immediate adverse effect on the trading price of our common stock.
Our business requires us to generate meaningful revenue from royalties and licensing agreements. To date, we have not recognized material amount of revenue from royalties for the commercial sale of drugs, and we do not know when we will be able to generate such meaningful revenue in the future.
Securities class action lawsuits or other litigation could result in substantial damages and may divert management’s time and attention from our business.
We have been subject to class action lawsuits in the past and we may be subject to lawsuits in the future, such as those that might occur if there was to be a change in our corporate strategy. These and other lawsuits are subject to inherent uncertainties, and the actual costs to be incurred relating to the lawsuit will depend upon many unknown factors. The outcome of litigation is necessarily uncertain, and we could be forced to expend significant resources in the defense of such suits, and we may not prevail. Monitoring and defending against legal actions is time-consuming for our management and detracts from our ability to fully focus our internal resources on our business activities. In addition, we may incur substantial legal fees and costs in connection with any such litigation. We have not established any reserves for any potential liability relating to any such potential lawsuits. It is possible that we could, in the future, incur judgments or enter into settlements of claims for monetary damages. A decision adverse to our interests in any such actions could result in the payment of substantial damages, or possibly fines, and could have an adverse effect on our cash flow, results of operations and financial position.
If our competitors develop technologies that are more effective than ours, our commercial opportunity will be reduced or eliminated.
The biotechnology and pharmaceutical industries are intensely competitive and subject to rapid and significant technological change. Many of the drugs that we are attempting to discover will be competing with existing therapies. In addition, a number of companies are pursuing the development of pharmaceuticals that target the same diseases and conditions that we are targeting. For example, the commercialization of new pharmaceutical products is highly competitive, and we face substantial competition with respect to our products in which there are existing therapies and drug candidates in development for the treatment of hematologic disorders and cancer that may be alternative therapies to our products. Many of our competitors, including a number of large pharmaceutical companies that compete directly with us, have significantly greater financial resources and expertise commercializing approved products than we do. Also, many of our competitors are large pharmaceutical companies that will have a greater ability to reduce prices for their competing drugs in an effort to gain market share and undermine the value proposition that we might otherwise be able to offer to payors. We face, and will continue to face, intense competition from pharmaceutical and biotechnology companies, as well as from academic and research institutions and government agencies, both in the US and abroad. Some of these competitors are pursuing the development of pharmaceuticals that target the same diseases and conditions as our research programs. Our competitors including fully integrated pharmaceutical companies have extensive drug discovery efforts and are developing novel small-molecule pharmaceuticals. We also face significant competition from organizations that are pursuing the same or similar technologies, including the discovery of targets that are useful in compound screening, as the technologies used by us in our drug discovery efforts.
Competition may also arise from:
•new or better methods of target identification or validation;
•generic versions of our products or of products with which we compete;
•other drug development technologies and methods of preventing or reducing the incidence of disease;
•new small molecules; or
•other classes of therapeutic agents.
Our competitors or their collaborative partners may utilize discovery technologies and techniques or partner with collaborators in order to develop products more rapidly or successfully than we or our collaborators are able to do. Many of our competitors, particularly large pharmaceutical companies, have substantially greater financial, technical and human resources and larger research and development staffs than we do. In addition, academic institutions, government agencies and other public and private organizations conducting research may seek patent protection with respect to potentially competitive products or technologies and may establish exclusive collaborative or licensing relationships with our competitors.
We believe that our ability to compete is dependent, in part, upon our ability to create, maintain and license scientifically-advanced technology and upon our and our collaborators’ ability to develop and commercialize
pharmaceutical products based on this technology, as well as our ability to attract and retain qualified personnel, obtain patent protection or otherwise develop proprietary technology or processes, secure effective market access by ensuring competitive pricing and reimbursement in territories of interest, and secure sufficient capital resources for the expected substantial time period between technological conception and commercial sales of products based upon our technology. The failure by any of our collaborators or us in any of those areas may prevent the successful commercialization of our potential drug targets.
Many of our competitors, either alone or together with their collaborative partners, have significantly greater experience than we do in:
•identifying and validating targets;
•screening compounds against targets; and
•undertaking preclinical testing and clinical trials.
Accordingly, our competitors may succeed in obtaining patent protection, identifying or validating new targets or developing new drug compounds before we do.
Our competitors might develop technologies and drugs that are more effective or less costly than any that are being developed by us or that would render our technology and product candidates obsolete and noncompetitive. In addition, our competitors may succeed in obtaining the approval of the FDA or other regulatory agencies for product candidates more rapidly. Companies that complete clinical trials, obtain required regulatory agency approvals and commence commercial sale of their drugs before us may achieve a significant competitive advantage, including certain patent and FDA marketing exclusivity rights that would delay or prevent our ability to market certain products. Any drugs resulting from our research and development efforts, or from our joint efforts with our existing or future collaborative partners, might not be able to compete successfully with competitors’ existing or future products or obtain regulatory approval in the US or elsewhere.
We face and will continue to face intense competition from other companies for collaborative arrangements with pharmaceutical and biotechnology companies, for establishing relationships with academic and research institutions and for licenses to additional technologies. These competitors, either alone or with their collaborative partners, may succeed in developing technologies or products that are more effective than ours.
Our ability to compete successfully will depend, in part, on our ability to:
•identify and validate targets;
•discover candidate drug compounds that interact with the targets we identify in a safe and efficacious way;
•attract and retain scientific and product development personnel;
•recruit subjects into our clinical trials;
•obtain and maintain required regulatory approvals;
•obtain patent or other proprietary protection for our new drug compounds and technologies;
•obtain access to manufacturing resources of sufficient standard and scale;
•enter commercialization agreements for our new drug compounds; and
•obtain and maintain appropriate reimbursement price and positive recommendations by HTA bodies.
Our stock price may be volatile, and our stockholders’ investment in our common stock could decline in value.
The market prices for our common stock and the securities of other biotechnology companies have been highly volatile and may continue to be highly volatile in the future. The following factors, in addition to other risk factors described in this section, may have a significant impact on the market price of our common stock:
•the progress and success of our clinical trials and preclinical activities (including studies and manufacture of materials) of our product candidates conducted by us;
•our ability to continue to sell our products in the US;
•our ability to enter into partnering opportunities across our pipeline;
•the receipt or failure to receive the additional funding necessary to conduct our business;
•selling of our common stock by large stockholders;
•presentations of detailed clinical trial data at medical and scientific conferences and investor perception thereof;
•announcements of technological innovations or new commercial products by our competitors or us;
•the announcement of regulatory applications, such as third party’s ANDA, seeking approval of generic versions of our marketed products;
•developments concerning proprietary rights, including patents;
•developments concerning our collaborations;
•publicity regarding actual or potential medical results relating to products under development by our competitors or us;
•regulatory developments in the US and foreign countries;
•changes in the structure of healthcare payment systems;
•litigation or arbitration;
•economic and other external factors or other disaster or crisis; and
•period-to-period fluctuations in financial results.
The UK’s withdrawal from the EU could adversely affect our business.*
The UK’s exit from the EU has created, and may continue to create, regulatory, trade and operational uncertainty. We may be required to obtain separate regulatory approvals in the UK and the EU, comply with differing regulatory requirements, and incur additional costs and administrative burdens in developing, manufacturing and commercializing our product candidates.
In addition, changes to trade arrangements between the UK and the EU may result in increased costs, delays or disruptions in the importation and exportation of our products and materials. Regulatory divergence between the UK and the EU may further complicate our operations and increase compliance risk.
Any of these factors could delay or prevent our ability to obtain approvals, limit our ability to commercialize our products in the UK or the EU, increase our operating costs, or otherwise materially and adversely affect our business, financial condition, results of operations and prospects.
If product liability lawsuits are successfully brought against us, we may incur substantial liabilities and may be required to limit commercialization of our products.
The testing and marketing of medical products and the sale of any products for which we obtain marketing approval exposes us to the risk of product liability claims. Product liability claims might be brought against us by consumers, healthcare providers, pharmaceutical companies or others selling or otherwise coming into contact with our products. If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit commercialization of our products. We carry product liability insurance that is limited in scope and amount and may not be adequate to fully protect us against product liability claims. If and when we obtain marketing approval for our product candidates, we intend to expand our insurance coverage to include the sale of commercial products; however, we may be unable to obtain product liability insurance on commercially reasonable terms or in adequate amounts. Our inability to obtain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the commercialization of pharmaceutical products we develop, alone or with corporate collaborators. We, or our corporate collaborators, might not be able to obtain insurance at a reasonable cost, if at all. While under various circumstances we are entitled to be indemnified against losses by our corporate collaborators, indemnification may not be available or adequate should any claim arise.
We depend on various scientific consultants and advisors for the success and continuation of our research and development efforts.
We work extensively with various scientific consultants and advisors. The potential success of our drug discovery and development programs depends, in part, on continued collaborations with certain of these consultants and advisors. We, and various members of our management and research staff, rely on certain of these consultants and advisors for expertise in our research, regulatory and clinical efforts. Our scientific advisors are not our employees and may have commitments to, or consulting or advisory contracts with, other entities that may limit their availability to us. We do not know if we will be able to maintain such consulting agreements or that such scientific advisors will not enter into consulting arrangements with competing pharmaceutical or biotechnology companies, any of which may have a detrimental impact on our research objectives and could have an adverse effect on our business, financial condition and results of operations.
While we have a strong compliance process in place to ensure we are complying with all requirements of law, our consulting or advisory contracts with our scientific consultants and advisors may be scrutinized under the Anti-Kickback Statute, the UK Bribery Act 2010, and other similar national and state-level legislation, which prohibit, among other things, companies from offering or paying anything of value as remuneration for ordering, purchasing, or recommending the ordering or purchasing of pharmaceutical and biological products that may be paid for, in whole or in part, by Medicare, Medicaid, or another federal healthcare program. Although there are several statutory exceptions and regulatory safe harbors that may protect these arrangements from prosecution or regulatory sanctions, our consulting and advising contracts may be subject to scrutiny if they do not fit squarely within an available exception or safe harbor.
If we use biological and hazardous materials in a manner that causes injury or violates laws, we may be liable for damages, penalties or fines.
Our research and development activities involve the controlled use of potentially harmful biological materials as well as hazardous materials, chemicals, animals, and various radioactive compounds. We cannot completely eliminate the risk of accidental contamination or injury from the use, storage, handling or disposal of these animals and materials. In the event of contamination or injury, we could be held liable for damages that result or for penalties or fines that may be imposed, and such liability could exceed our resources. We are also subject to federal, state and local laws and regulations governing the use, storage, handling and disposal of these materials and specified waste products. The cost of compliance with, or any potential violation of, these laws and regulations could be significant.
Our information technology systems, or those used by our CROs or other contractors or consultants, may fail or suffer other breakdowns, cyber-attacks, or information security breaches.
We are dependent upon information technology systems, infrastructure, and data to operate our business. While we believe our cybersecurity measures are adequate, our cybersecurity risk management, strategy and governance may be found to be inadequate that could harm our business. We rely on third-party vendors and their information technology systems. Despite the implementation of security measures, our recovery systems, security protocols, network protection mechanisms and other security measures and those of our CROs and other contractors and consultants are vulnerable to
compromise from natural disasters; terrorism; war; telecommunication and electric failures; traditional computer hackers; malicious code (such as computer viruses or worms); employee error, theft or misuse; denial-of-service attacks; cyber-attacks by sophisticated nation-state and nation-state supported actors including ransomware; or other system disruptions. We receive, generate and store significant and increasing volumes of personal (including health), confidential and proprietary information. There can be no assurance that we, or our collaborators, CROs, third-party vendors, contractors and consultants will be successful in efforts to detect, prevent, protect against or fully recover systems or data from all breakdowns, service interruptions, attacks or breaches. Any breakdown, cyber-attack or information security breach could result in a disruption of our drug development programs or other aspects of our business. For example, the loss of clinical trial data from completed or ongoing clinical trials for a product candidate could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach were to result in a loss of or damage to our data or applications, or inappropriate disclosure of personal, confidential or proprietary information, we could incur liability, incur significant remediation or litigation costs, result in product development delays, disrupt key business operations, cause loss of revenue and divert attention of management and key information technology resources.
Hackers and data thieves are increasingly sophisticated and operate large-scale and complex automated attacks, including on companies within the healthcare industry. As the cyber-threat landscape evolves, these threats are likely growing in frequency, sophistication and intensity and are increasingly difficult to detect. The costs of maintaining or upgrading our cyber-security systems at the level necessary to keep up with our expanding operations and prevent against potential attacks are increasing. Cyber threats may be generic, or they may be targeted against our information systems. Our network and storage applications and those of our contract manufacturing organizations, collaborators, contractors, CROs or vendors may be subject to unauthorized access or processing by hackers or breached due to operator or other human error, theft, malfeasance or other system disruptions. We may be unable to anticipate or immediately detect information security incidents and the damage caused by such incidents. These data breaches and any unauthorized access, processing or disclosure of our information or intellectual property could compromise our intellectual property and expose our sensitive business information. Such attacks, such as in the case of a ransomware attack, also may interfere with our ability to continue to operate and may result in delays and shortcomings due to an attack that may encrypt our or our service providers’ or partners’ systems unusable. Additionally, because our services involve the processing of personal information and other sensitive information about individuals, we are subject to various laws, regulations, industry standards, and contractual requirements related to such processing. Any event that leads to unauthorized access, processing or disclosure of personal information, including personal information regarding our clinical trial participants or employees, could harm our reputation and business, compel us to comply with federal and/or state breach notification laws and foreign law equivalents, subject us to investigations and mandatory corrective action, and otherwise subject us to liability under laws, regulations or contracts that protect the privacy and security of personal information, which could disrupt our business, damage our reputation with our stakeholders, result in increased costs or loss of revenue, lead to negative publicity or result in significant financial exposure. The California Consumer Privacy Act of 2018 (CCPA), in particular, includes a private right of action for California consumers whose personal information is impacted by a data security incident resulting from a company’s failure to maintain reasonable security procedures, and hence may result in civil litigation in the event of a security breach impacting such information. In addition, legislators and regulators in the US have enacted and are proposing new and more robust privacy and cybersecurity laws and regulations in response to increasing broad-based cyberattacks, including the CCPA and New York SHIELD Act. Notably, on July 26, 2023, the SEC adopted a final rule on cybersecurity risk management, strategy, governance and incident disclosure (SEC Cyber Rule). The SEC Cyber Rule requires public companies to make current disclosures about material cybersecurity incidents as well as annual disclosures of material information about their cybersecurity risk management, strategy and governance. The SEC Cyber Rule became effective on September 5, 2023. New data security laws add additional complexity, requirements, restrictions and potential legal risk, and compliance programs may require additional investment in resources, and could impact strategies and availability of previously useful data. Failure to timely identify and disclose a material cybersecurity incident could result in SEC enforcement actions, litigation, and reputational damage.
The costs to respond to a security breach and/or to mitigate any identified security vulnerabilities could be significant, our efforts to address these issues may not be successful, and these issues could result in interruptions, delays, negative publicity, loss of customer trust, and other harms to our business and competitive position. Remediation of any potential security breach may involve significant time, resources, and expenses. We could be required to fundamentally change our business activities and practices in response to a security breach and our systems or networks may be perceived as less desirable, which could negatively affect our business and damage our reputation.
A security breach may cause us to breach our contracts with third parties. Our agreements with relevant stakeholders such as collaborators may require us to use legally required, industry-standard or reasonable measures to
safeguard personal information. A security breach could lead to claims by relevant stakeholders that we have failed to comply with such contractual obligations, or require us to cooperate with these stakeholders in their own compliance efforts related to the security breach. In addition, any non-compliance with our data privacy obligations in our contracts or our inability to flow down such obligations from relevant stakeholders to our vendors may cause us to breach our contracts. As a result, we could be subject to legal action or the relevant stakeholders could end their relationships with us. There can be no assurance that the limitations of liability in our contracts would be enforceable or adequate or would otherwise protect us from liabilities or damages.
We may not have adequate insurance coverage for security incidents or breaches. The successful assertion of one or more large claims against us that exceeds our available insurance coverage, or results in changes to our insurance policies (including premium increases or the imposition of large deductible or co-insurance requirements), could have an adverse effect on our business. In addition, we cannot be sure that our existing insurance coverage will continue to be available on acceptable terms or that our insurers will not deny coverage as to any future claim.
Future equity issuances or a sale of a substantial number of shares of our common stock may cause the price of our common stock to decline.
Because we will continue to need additional capital in the future to continue to expand our business, we may conduct additional equity offerings. We have an Open Market Sale Agreement with Jefferies entered on August 4, 2020, and amended and restated on August 2, 2024, pursuant to which, we may sell from time to time, through Jefferies, shares of our common stock in sales deemed to be “at-the-market offerings” as defined in Rule 415 under the Securities Act, subject to conditions specified in the Open Market Sale Agreement. On August 2, 2024, we filed a shelf registration statement with the SEC to register the offering, issuance and sale by us of up to $250.0 million in the aggregate of securities identified from time to time in one or more offerings, including up to $100.0 million of shares of our common stock that may be offered, issued and sold under the Open Market Sale Agreement. As of March 31, 2026, we have not sold any shares of common stock under the Open Market Sale Agreement.
We may also in the future enter into underwriting or sales agreements with financial institutions for the offer and sale of any combination of common stock, preferred stock, debt securities and warrants in one or more offerings. If we or our stockholders sell, or if it is perceived that we or they will sell, substantial amounts of our common stock in the public market, the market price of our common stock could fall. A decline in the market price of our common stock could make it more difficult for us to sell equity or equity-related securities in the future at a time and price that we deem appropriate. In addition, future sales by us of our common stock may be dilutive to existing stockholders. Furthermore, if we obtain funds through a credit facility or through the issuance of debt or preferred securities, these securities would likely have rights senior to the rights of our common stockholders, which could impair the value of our common stock.
Risks Related to Clinical Development and Regulatory Approval
Enacted or future legislation, and/or potentially unfavorable pricing regulations or other healthcare reform initiatives, may increase the difficulty and cost for us to obtain regulatory approval of our product candidates and/or commercialize our products or our product candidates, once approved, and affect the prices we may set or obtain.*
The regulations that govern, among other things, regulatory approvals, coverage, pricing and reimbursement for new drug products vary widely from country to country. In the US and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay regulatory approval of our product candidates, restrict or regulate post-approval activities and affect our ability to successfully sell our products, or any product candidates for which we obtain regulatory approval in the future. In particular, in March 2010, the Affordable Care Act was enacted, which substantially changed the way healthcare is financed by both governmental and private insurers, and continues to significantly impact the US pharmaceutical industry. On June 17, 2021, the US Supreme Court dismissed a legal challenge to the Affordable Care Act brought by several states (which argued that, without the individual mandate, the entire Affordable Care Act was unconstitutional) without specifically ruling on the constitutionality of the law. It is unclear how future actions before the Supreme Court, other such litigation, and the healthcare reform measures of future presidential administrations will impact the Affordable Care Act and our business.
There have been, and likely will continue to be, legislative and regulatory proposals at the foreign, federal and state levels directed at broadening the availability of healthcare and containing or lowering the cost of healthcare. We cannot predict the initiatives that may be adopted in the future. The continuing efforts of the government, insurance
companies, managed care organizations and other payors of healthcare services to contain or reduce the costs of healthcare and/or impose price controls may adversely affect, for example:
•the demand for our products, or our product candidates, if we obtain regulatory approval;
•our ability to set a price that we believe is fair for our products;
•our ability to generate revenue and achieve or maintain profitability;
•the level of taxes that we are required to pay; and
•the availability of capital.
Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors, which may adversely affect our future profitability.
In the US, the EU and other potentially significant markets for our current and future products, government authorities and third-party payors are increasingly attempting to limit or regulate the price of medical products and services, particularly for new and innovative products and therapies, which has resulted in lower average selling prices. In the US, there have been several Congressional inquiries and federal legislation designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer-sponsored patient assistance programs, and reform government program reimbursement methodologies for drugs.
Recent healthcare reform measures, including the Inflation Reduction Act and changes to government healthcare programs, could adversely affect our business. U.S. federal and state healthcare reforms have resulted in, and are expected to continue to result in, significant changes to the pricing, reimbursement and coverage of pharmaceutical products. For example, the Inflation Reduction Act (IRA) allows Medicare to negotiate prices for certain drugs, imposes inflation-based rebates for products covered under Medicare Parts B and D, and redesigns the Medicare Part D benefit in ways that increase financial obligations for manufacturers, while the elimination of the Medicaid drug rebate cap may increase our rebate liability. Additionally, OBBBA broadened the scope of IRA’s exclusion of certain orphan drugs from price negotiations. These and other reforms may reduce the prices we can charge for our products, increase our rebate and discount obligations, and negatively affect reimbursement, which could adversely impact our revenues, margins and operations. The full impact of these measures, as well as potential future legislative, regulatory or judicial developments, remains uncertain and could materially and adversely affect our business, financial condition, results of operations and prospects.
Other proposed administrative actions may affect our government pricing responsibilities. For example, there are pending legal and legislative developments relating to the 340B Drug Pricing Program, including ongoing litigation challenging federal enforcement actions against manufacturers and recently introduced and enacted state legislation. It remains to be seen how these drug pricing initiatives will affect the broader pharmaceutical industry. Although none of our products are currently subject to Medicare price negotiation under the IRA, the program is expected to expand to additional drugs in the future years. If any of our products were selected for negotiation, the negotiated prices could significantly lower than the prices we currently receive, which could materially reduce our revenues and profitability.
The current presidential administration has also signaled its intent to pursue healthcare reform measures, including those aimed at reducing prescription drug prices. For example, President Trump has signed multiple executive orders addressing prescription drug pricing and access, including: an order on May 12, 2025 aiming to establish a most favored nation (MFN) drug pricing policy that would tie US drug prices to the prices paid for drugs in other countries. Since the May 12, 2025 MFN executive order, the Trump administration has continued to exert pressure on drug manufacturers to implement MFN pricing. Over a dozen large pharmaceutical manufacturers have entered into agreements with the Trump Administration to offer lower prices for their drugs. Additionally, CMS has taken action to implement the administration’s MFN pricing policy, including by announcing a new voluntary payment model where drug manufacturers may offer supplemental rebates to participating state Medicaid programs to provide such Medicaid programs with a “most favored nation” price for participating manufacturers’ products, as well proposing mandatory payment models where, if finalized, manufacturers of certain Medicare Part B and Medicare Part D drugs would be assessed rebates if the prices for such products exceed those paid in economically comparable countries. The Trump administration also announced the launch of a new direct-to-consumer website in February 2026 that is intended to make certain drugs available to consumers at significant discounts. It remains to be seen how these drug pricing initiatives will affect the broader pharmaceutical industry.
At the state level, individual states are increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing. Specifically, several US states and localities have enacted legislation requiring pharmaceutical companies to establish marketing compliance programs, file periodic reports, and/or make periodic public disclosures on sales, marketing, pricing, clinical trials, and other activities. Other state laws prohibit certain marketing-related activities, including the provision of gifts, meals or other items to certain healthcare providers, and restrict the ability of manufacturers to offer co-pay support to patients for certain prescription drugs. Several state laws require disclosures related to state agencies and/or commercial purchasers with respect to price increases and new product launches that exceed certain thresholds as identified in the relevant statutes. Some of these laws and regulations contain ambiguous requirements that government officials have not yet clarified. Given the lack of clarity in the laws and their implementation, our reporting actions could be subject to the penalty provisions of the pertinent state laws and regulations. Another emerging trend at the state level is the establishment of prescription drug affordability boards, some of which will prospectively permit certain states to establish upper payment limits for drugs that the state has determined to be “high-cost.” Prescription drug affordability boards in several states have begun identifying products for affordability reviews and issuing information requests to manufacturers to determine whether upper payment limits may be justified.
Furthermore, the increased emphasis on managed healthcare in the US and on country and regional pricing and reimbursement controls in the EU and the UK will put additional pressure on product pricing, reimbursement and usage, which may adversely affect our sales and results of operations. These pressures can arise from rules and practices of managed care groups, judicial decisions and governmental laws and regulations related to Medicare, Medicaid, healthcare reform, pharmaceutical reimbursement policies and pricing in general. Legislative and regulatory proposals have been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products.
We cannot predict the likelihood, nature, or extent of health reform initiatives that may arise from future legislation or administrative action. However, we expect these initiatives to increase pressure on drug pricing. Further, certain broader legislation that is not targeted to the healthcare industry may nonetheless adversely affect our profitability. If we or any third parties we may engage are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we or such third parties are not able to maintain regulatory compliance, our product candidates may lose any regulatory approval that may have been obtained and we may not achieve or sustain profitability.
See “Business – Government Regulation – Healthcare Reform” contained in Part I, Item 1 of our Annual Report on Form 10-K for the year ended December 31, 2025, for additional information.
Regulatory approval for any approved product is limited by the FDA, the EC and other regulators to those specific indications and conditions for which clinical safety and efficacy have been demonstrated, and we may incur significant liability if it is determined that we are promoting the “off-label” use of our products or any of our future product candidates if approved.
Any regulatory approval is limited to those specific diseases, indications and patient populations for which a product is deemed to be safe and effective by the FDA, the EC and other regulators. For example, the FDA-approved label for TAVALISSE is only approved for use in adults with ITP who have had an insufficient response to other treatments and for REZLIDHIA is only approved for use in adult patients with R/R AML with a susceptible IDH1 mutation as detected by an FDA-approved test. Further, GAVRETO is approved by the FDA for the treatment of adult patients with metastatic RET fusion-positive NSCLC and has a conditional approval for the treatment of adult and pediatric patients 12 years of age and older with advanced or metastatic RET fusion-positive thyroid cancer. In addition to the FDA approval required for new formulations, any new indication for an approved product also requires FDA approval. If we are not able to obtain FDA approval for any desired future indications for our products and product candidates, or if we are not able to maintain a conditional approval or transition a conditional approval to full approval, our ability to effectively market and sell our products may be reduced and our business may be adversely affected.
While physicians may choose to prescribe drugs for uses that are not described in the product’s labeling and for uses that differ from those tested in clinical studies and approved by the regulatory authorities, our ability to promote the products is limited to those indications and patient populations that are specifically approved by the FDA. These “off-label” uses are common across medical specialties and may constitute an appropriate treatment for some patients in varied circumstances. We have implemented compliance and monitoring policies and procedures, including a process for internal review of promotional materials, to deter the promotion of our products for off-label uses. We cannot guarantee that these compliance activities will prevent or timely detect off-label promotion by sales representatives or other personnel in their communications with healthcare professionals, patients and others, particularly if these activities are concealed from us. Regulatory authorities in the US generally do not regulate the behavior of physicians in their choice of treatments. Regulatory authorities do, however, restrict communications by pharmaceutical companies on the subject of off-label use.
If our promotional activities fail to comply with the FDA’s or other competent national authority’s regulations or guidelines, we may be subject to warnings from, or enforcement action by, these regulatory authorities. In addition, our failure to follow FDA rules and guidelines relating to promotion and advertising may cause the FDA to issue warning letters or untitled letters, suspend or withdraw an approved product from the market, require a recall or institute fines, which could result in the disgorgement of money, operating restrictions, injunctions or civil or criminal enforcement, and other consequences, any of which could harm our business.
Notwithstanding the regulatory restrictions on off-label promotion, the FDA and other regulatory authorities allow companies to engage in truthful, non-misleading and non-promotional scientific exchange concerning their products. We engage in medical education activities and communicate with investigators and potential investigators regarding our clinical trials. If the FDA or other regulatory or enforcement authorities determine that our communications regarding our marketed product are not in compliance with the relevant regulatory requirements and that we have improperly promoted off-label uses, or that our communications regarding our investigational products are not in compliance with the relevant regulatory requirements and that we have improperly engaged in pre-approval promotion, we may be subject to significant liability, including civil and administrative remedies as well as criminal sanctions.
Delays in clinical testing could result in increased costs to us.
We may not be able to initiate or continue clinical studies or trials for our product candidates if we are unable to locate and enroll a sufficient number of eligible patients to participate in these clinical trials as required by the FDA or other regulatory authorities, whether due to the impacts of a global pandemic, global geopolitical tensions or otherwise. Even if we are able to enroll a sufficient number of patients in our clinical trials, if the pace of enrollment is slower than we expect, the development costs for our product candidates may increase and the completion of our clinical trials may be delayed, or our clinical trials could become too expensive to complete. Significant delays in clinical testing could negatively impact our product development costs and timing. Our estimates regarding timing are based on a number of assumptions, including assumptions based on past experience with our other clinical programs. If we are unable to enroll the patients in these trials at the projected rate, the completion of the clinical program could be delayed and the costs of conducting the program could increase, either of which could harm our business.
Clinical trials can be delayed for a variety of reasons, including delays in obtaining regulatory approval to commence a study, delays from scaling up of a study, delays in reaching agreement on acceptable clinical trial agreement terms with prospective clinical sites, delays in obtaining institutional review board approval to conduct a study at a prospective clinical site or delays in recruiting subjects to participate in a study. In addition, we typically rely on third-party clinical investigators to conduct our clinical trials and other third-party organizations to oversee the operations of such trials and to perform data collection and analysis. The clinical investigators are not our employees, and we cannot control the amount or timing of resources that they devote to our programs. Failure of the third-party organizations to meet their obligations, whether due to the potential future impacts of a global pandemic, or global geopolitical tensions, could adversely affect clinical development of our products. As a result, we may face additional delaying factors outside our control if these parties do not perform their obligations in a timely fashion. For example, any number of those issues could arise with our clinical trials causing a delay. Delays of this sort could occur for the reasons identified above or other reasons. If we have delays in conducting the clinical trials or obtaining regulatory approvals, our product development costs will increase. For example, we may need to make additional payments to third-party investigators and organizations to retain their services or we may need to pay recruitment incentives. If the delays are significant, our financial results and the commercial prospects for our product candidates will be harmed, and our ability to become profitable will be delayed. Moreover, these third-party investigators and organizations may also have relationships with other commercial entities, some of which may compete with us. If these third-party investigators and organizations assist our competitors at our expense, it could harm our competitive position.
We have conducted in the past and are currently conducting or may conduct in the future clinical trials in the US and outside the US. In the past, we had clinical trial sites in Russia and Ukraine for our wAIHA trial, which trials have concluded. Recent actions taken by the Russian Federation in Ukraine and surrounding areas have destabilized the region and caused the adoption of comprehensive sanctions by, among others, the EU, the US and the UK, which restrict a wide range of trade and financial dealings with Russia and Russian persons, as well as certain regions in Ukraine. Also, recent global tensions, conflicts, and wars may result in disruptions in the broader global economic environment. Further, some patients may not be able to comply with clinical trial protocols if the conflict impedes patient movement or interrupts healthcare services. In addition, clinical trial site initiation and patient enrollment may be delayed, and we may not be able to access sites for initiation and monitoring in regions affected by the global geopolitical tensions, including due to the prioritization of hospital resources away from clinical trials or as a result of warfare, violence, government-imposed
curfews, or events or other governmental actions that restrict movement. We could also experience disruptions in our supply chain or limits our ability to obtain sufficient materials for our drug products in certain regions.
A Fast Track designation by the FDA may not lead to a faster development or regulatory review and does not increase the likelihood that our product candidates will receive approval.
We may seek fast track designation for our product candidates. If a product is intended for the treatment of a serious or life-threatening disease or condition and it demonstrates the potential to address unmet medical needs for such a disease or condition, the sponsor may apply for FDA fast track designation for a particular indication. We may seek fast track designation for our product candidates, but there is no assurance that the FDA will grant this designation to any of our proposed product candidates, even if such a designation has been granted to similar products. Marketing applications submitted by sponsors of products in fast track development may qualify for priority review under the policies and procedures offered by the FDA, but the fast track designation does not assure any such qualification or ultimate marketing approval by the FDA.
The FDA has broad discretion whether or not to grant fast track designation, so even if we believe a particular product candidate is eligible for this designation, there can be no assurance that the FDA would decide to grant it. Even if we do receive fast track designation, we may not experience a faster development process, review or approval compared to conventional FDA procedures or pathways and receiving a fast track designation does not provide assurance of ultimate FDA approval. In addition, the FDA may withdraw fast track designation at any time, including if it believes that the designation is no longer supported by data from our clinical development program.
Public perception of the risk-benefit balance for our product candidates may be affected by adverse events in clinical trials involving our product candidate or other treatments.
Negative perception of the efficacy, safety, or tolerability of any investigational medicines that we develop, or of other products similar to products we are developing, could adversely affect our ability to conduct our business, advance our investigational medicines, or obtain regulatory approvals.
Adverse events in clinical trials of our investigational medicines or in clinical trials of others developing similar products could result in a decrease in the perceived benefit of one or more of our programs, increased regulatory scrutiny, decreased confidence by patients and clinical trial collaborators in our investigational medicines, and less demand for any product that we may develop. If and when they are used in clinical trials, our developmental candidates and investigational medicines could result in a greater quantity of reportable adverse events, including suspected unexpected serious adverse reactions, other reportable negative clinical outcomes, manufacturing reportable events or material clinical events that could lead to clinical delay or hold by the FDA or applicable regulatory authority or other clinical delays, any of which could negatively impact the perception of one or more of our programs, as well as our business as a whole. In addition, responses by US, state, or foreign governments to negative public perception may result in new legislation or regulations that could limit our ability to develop any investigational medicines or commercialize any approved products, obtain or maintain regulatory approval, or otherwise achieve profitability. More restrictive statutory regimes, government regulations, or negative public opinion would have an adverse effect on our business, financial condition, results of operations, and prospects and may delay or impair the development of our investigational medicines and commercialization of any approved products or demand for any products we may develop.
We lack the capability to manufacture compounds for clinical development, and we rely on and intend to continue relying on third parties for commercial supply, manufacturing and distribution, if any, of our product candidates which receive regulatory approval and we may be unable to obtain required material or product in a timely manner, at an acceptable cost or at a quality level required to receive regulatory approval.
We currently do not have the manufacturing capabilities or experience necessary to produce our products or any product candidates for clinical trials. We currently use three API manufacturing facilities and three finished goods manufacturing facilities for our products. We do not currently have, nor do we plan to acquire the infrastructure or capability to supply, manufacture or distribute preclinical, clinical or commercial quantities of drug substances or products. For each clinical trial of our unpartnered product candidates, we rely on third-party manufacturers for the APIs, as well as various manufacturers to manufacture starting components, excipients and formulated drug products. Our ability to develop our product candidates, and our ability to commercially supply our products will depend, in part, on our ability to successfully obtain the APIs and other substances and materials used in our product candidates from third parties and to have finished products manufactured by third parties in accordance with regulatory requirements and in sufficient
quantities for preclinical and clinical testing and commercialization. If we fail to develop and maintain supply relationships with these third parties, we may be unable to continue to develop or commercialize our product candidates.
Since our commercialization, we have sold inventory quantities that were acquired or produced before FDA approval and therefore did not reflect full production costs, as pre-approval manufacturing costs were previously expensed to research and development. Specifically, we utilized zero-cost API inventory for TAVALISSE, which reduced cost of product sales in those periods. As post-approval inventory is acquired or produced, inventory and cost of product sales reflect the full manufacturing cost. Further, the imposition or threat of imposition of trade policies, tariffs (including retaliatory tariffs), taxes and other cross-border operations could also result in higher cost of product sales.
We rely and will continue to rely on certain third parties, including those located outside the US, as our limited source suppliers of certain materials and finished products. In the ordinary course of business, we enter into agreements with contract manufacturers to manufacture our inventory products. For example, in October 2024, we entered into an agreement with a third-party contract manufacturer to manufacture TAVALISSE that is expected to be delivered starting in fiscal year 2026 through 2029. Although the agreement provides a cancellation clause with or without cause upon written notice, we may or may not be subject to payment of cancellation fees. The level of cancellation fees is generally dependent on the timing of the written notice in relation to the commencement date of work, with the maximum cancellation fees equal to the full price of the work order. The drug substances and other materials used in our product candidates are currently available only from one or a limited number of suppliers or manufacturers and certain of our finished product candidates are manufactured by one or a limited number of contract manufacturers. Any of these existing suppliers or manufacturers may:
•fail to supply us with product on a timely basis or in the requested amount due to unexpected damage to or destruction of facilities or equipment or otherwise;
•fail to increase manufacturing capacity and produce drug product and components in larger quantities and at higher yields in a timely or cost-effective manner, or at all, to sufficiently meet our commercial needs;
•be unable to meet our production demands due to issues related to their reliance on sole-source suppliers and manufacturers;
•supply us with product that fails to meet regulatory requirements;
•become unavailable through business interruption or financial insolvency;
•lose regulatory status as an approved source;
•be unable or unwilling to renew current supply agreements when such agreements expire on a timely basis, on acceptable terms or at all; or
•discontinue production or manufacturing of necessary drug substances or products.
Our current and anticipated future dependence upon these third-party manufacturers may adversely affect our ability to develop and commercialize product candidates on a timely and competitive basis, which could have an adverse effect on sales, results of operations and financial condition. If we were required to transfer manufacturing processes to other third-party manufacturers and we were able to identify an alternative manufacturer, we would still need to satisfy various regulatory requirements. Satisfaction of these requirements could cause us to experience significant delays in receiving an adequate supply of our products and products in development and could be costly. Moreover, we may not be able to transfer processes that are proprietary to the manufacturer, if any. These manufacturers may not be able to produce material on a timely basis or manufacture material at the quality level or in the quantity required to meet our development timelines and applicable regulatory requirements and may also experience a shortage in qualified personnel. Certain of our third-party manufacturers are located outside of the US, and import materials from other countries including China to produce our products. The tensions between the US and other countries including China have led to a series of tariffs and sanctions being imposed on imports by the US, as well as other business restrictions. In response, other countries, notably China, have threatened or imposed tariffs or other trade sanctions on products manufactured in the US. Geopolitical developments, including changes arising as a result of the 2024 US presidential election, may lead to further developments with respect to the imposition or threat of imposition of trade policies, tariffs (including retaliatory tariffs), taxes and other limitations on cross-border operations. Such tensions could adversely impact us and our third-party manufacturers. We may not be able to maintain or renew our existing third-party manufacturing arrangements, or enter into new arrangements, on acceptable terms, or at all. Our third-party manufacturers could terminate or decline to renew our manufacturing arrangements based on their own business priorities, at a time that is costly or inconvenient for us. If we are unable to
contract for the production of materials in sufficient quantity and of sufficient quality on acceptable terms, our planned clinical trials may be significantly delayed. Manufacturing delays could postpone the filing of our investigational new drug (IND) applications and/or the initiation or completion of clinical trials that we have currently planned or may plan in the future.
Drug manufacturers are subject to ongoing periodic unannounced inspection by the FDA, the Drug Enforcement Administration, the EMA, national competent authorities in the EU and UK and other federal and state government and regulatory agencies to ensure strict compliance with cGMP and other government regulations and corresponding foreign standards. We do not have control over third-party manufacturers’ compliance with these regulations and standards and they may not be able to comply. Switching manufacturers may be difficult because the number of potential manufacturers is limited. It may be difficult or impossible for us to find a replacement manufacturer quickly on acceptable terms, or at all. Additionally, if we are required to enter into new supply arrangements, we may not be able to obtain approval from the FDA of any alternate supplier in a timely manner, or at all, which could delay or prevent the clinical development and commercialization of any related product candidates. Failure of our third-party manufacturers or us to comply with applicable regulations, whether due to the impacts of a global pandemic or otherwise, could result in sanctions being imposed on us, including fines, civil penalties, delays in or failure to grant marketing approval of our product candidates, injunctions, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of products and compounds, operating restrictions and criminal prosecutions, warning or similar letters or civil, criminal or administrative sanctions against us, any of which could adversely affect our business.
Any product for which we have obtained regulatory approval, or for which we obtain approval in the future, is subject to, or will be subject to, extensive ongoing regulatory requirements by the FDA, EMA, MHRA and other comparable regulatory authorities, and if we fail to comply with regulatory requirements or if we experience unanticipated problems with our products, we may be subject to penalties, we may be unable to generate revenue from the sale of such products, our potential for generating positive cash flow will be diminished, and the capital necessary to fund our operations will be increased.*
We commercialize our products in the US and we have entered into commercialization agreements with third parties to commercialize our products outside the US. Any product for which we have obtained regulatory approval, or for which we obtain regulatory approval in the future, along with the manufacturing processes and practices, post-approval clinical research, product labeling, advertising and promotional activities for such product, are subject to continual requirements of, and review by, the FDA, the European Medicines Agency (EMA) and other comparable international regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports, registration and listing requirements, cGMP requirements relating to manufacturing, quality control, quality assurance and corresponding maintenance of records and documents, requirements regarding the distribution of samples to physicians, import and export requirements and recordkeeping. If we or our suppliers encounter manufacturing, quality or compliance difficulties with respect to our products or any of our product candidates, when and if approved, whether due to the impacts of a global pandemic (including as a result of disruptions of global shipping and the transport of products) or otherwise, we may be unable to obtain or maintain regulatory approval or meet commercial demand for such products, which could adversely affect our business, financial conditions, results of operations and growth prospects.
Promotional communications with respect to prescription drugs are subject to a variety of legal and regulatory restrictions and must be consistent with the information in the product’s approved labeling. Thus, we will not be able to promote any products we develop for indications or uses for which they are not approved.
In addition, the FDA often requires post-marketing testing and surveillance to monitor the effects of products. The FDA, the EMA and other comparable international regulatory agencies may condition approval of our product candidates on the completion of such post-marketing clinical studies. These post-marketing studies may suggest that a product causes undesirable side effects or may present a risk to the patient. Additionally, the FDA may require REMS to help ensure that the benefits of the drug outweigh its risks. A REMS may be required to include various elements, such as a medication guide or patient package insert, a communication plan to educate healthcare providers of the drug’s risks, limitations on who may prescribe or dispense the drug, requirements that patients enroll in a registry or undergo certain health evaluations or other measures that the FDA deems necessary to ensure the safe use of the drug. Additionally, approval of a drug under an accelerated drug approval program may be withdrawn or the labeled indication of the drug changed if trials fail to verify clinical benefit or do not demonstrate sufficient clinical benefit to justify the risks associated with the drug. For example, GAVRETO is approved under accelerated approval for the treatment of adult and pediatric patients 12 years of age and older with advanced or metastatic RET fusion-positive thyroid cancer. We have satisfied our post-marketing commitment with respect to the AcceleRET-Lung study for the NSCLC indication; however, discussions with the FDA regarding confirmatory requirements for the thyroid cancer indication remain ongoing. The FDA has demonstrated an increased
willingness to withdraw accelerated approvals where confirmatory trials have not been completed or have failed to verify clinical benefit. If we are unable to satisfy the FDA’s confirmatory requirements for the thyroid cancer indication, the FDA could withdraw approval for that indication, which would reduce the addressable patient population for GAVRETO and could adversely affect our revenues and commercial strategy.
Discovery after approval of previously unknown problems with any of our products, manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in actions such as:
•restrictions on our ability to conduct clinical trials, including full or partial clinical holds on ongoing or planned trials;
•restrictions on product manufacturing processes;
•restrictions on the marketing of a product;
•restrictions on product distribution;
•requirements to conduct post-marketing clinical trials;
•untitled or warning letters or other adverse publicity;
•withdrawal of products from the market;
•refusal to approve pending applications or supplements to approved applications that we submit;
•recall of products;
•refusal to permit the import or export of our products;
•product seizure;
•fines, restitution or disgorgement of profits or revenue;
•refusal to allow us to enter into supply contracts, including government contracts;
•injunctions; or
•imposition of civil or criminal penalties.
If such regulatory actions are taken, the value of our company and our operating results will be adversely affected. Additionally, if the FDA, the EMA or any other comparable international regulatory agency withdraws its approval of a product that is or may be approved, we will be unable to generate revenue from the sale of that product in the relevant jurisdiction, our potential for generating positive cash flow will be diminished and the capital necessary to fund our operations will be increased. Accordingly, we continue to expend significant time, money and effort in all areas of regulatory compliance, including manufacturing, production, product surveillance, post-marketing studies and quality control.
If any of our third-party contractors fail to perform their responsibilities to comply with FDA rules and regulations, the marketing and sales of our products could be delayed and we may be subject to enforcement action, which could decrease our revenues.
Conducting our business requires us to manage relationships with third-party contractors. As a result, our success depends partially on the success of these third parties in performing their responsibilities to comply with FDA rules and regulations. Although we pre-qualify our contractors and we believe that they are fully capable of performing their contractual obligations, we cannot directly control the adequacy and timeliness of the resources and expertise that they apply to these activities.
If any of our partners or contractors fail to perform their obligations in an adequate and timely manner, or fail to comply with the FDA’s rules and regulations, then the marketing and sales of our products could be delayed. The FDA may also take enforcement actions against us based on compliance issues identified with our contractors. If any of these events occur, we may incur significant liabilities, which could decrease our revenues. For example, sales and medical science liaison or MSL personnel, including contractors, must comply with FDA requirements for the advertisement and promotion of products.
If we are unable to obtain regulatory approval to market products in the US and foreign jurisdictions, we will not be permitted to commercialize products we or our collaborative partners may develop.
We cannot predict whether regulatory clearance will be obtained for any product that we, or our collaborative partners, hope to develop. Satisfaction of regulatory requirements typically takes many years, is dependent upon the type, complexity and novelty of the product and requires the expenditure of substantial resources. Of particular significance to us are the requirements relating to research and development and testing.
Before commencing clinical trials in humans in the US, we, or our collaborative partners, will need to submit and receive approval from the FDA of an IND application. Clinical trials are subject to oversight by institutional review boards and the FDA and:
•must be conducted in conformance with the FDA’s good clinical practices and other applicable regulations;
•must meet requirements for institutional review board oversight;
•must meet requirements for informed consent;
•are subject to continuing FDA and regulatory oversight;
•may require large numbers of test subjects; and
•may be suspended by us, our collaborators or the FDA at any time if it is believed that the subjects participating in these trials are being exposed to unacceptable health risks or if the FDA finds deficiencies in the IND or the conduct of these trials.
While we have stated that we intend to file additional INDs for future product candidates, this is only a statement of intent, and we may not be able to do so because we may not be able to identify potential product candidates. In addition, the FDA may not approve any IND we or our collaborative partners may submit in a timely manner, or at all.
Before receiving FDA approval to market a product, we must demonstrate with substantial clinical evidence that the product is safe and effective in the patient population and the indication that will be treated. Data obtained from preclinical and clinical activities are susceptible to varying interpretations that could delay, limit or prevent regulatory approvals. In addition, delays or rejections may be encountered based upon additional government regulation from future legislation or administrative action or changes in FDA policy during the period of product development, clinical trials and FDA regulatory review. Failure to comply with applicable FDA or other applicable regulatory requirements may result in criminal prosecution, civil penalties, recall or seizure of products, total or partial suspension of production or injunction, adverse publicity, as well as other regulatory action against our potential products or us. Additionally, we have limited experience in conducting and managing the clinical trials necessary to obtain regulatory approval.
If regulatory approval of a product is granted, this approval will be limited to those indications or disease states and conditions for which the product is demonstrated through clinical trials to be safe and efficacious. We cannot assure that any compound developed by us, alone or with others, will prove to be safe and efficacious in clinical trials and will meet all of the applicable regulatory requirements needed to receive marketing approval.
Outside the US, our ability, or that of our collaborative partners, to market a product is contingent upon receiving a marketing authorization from the appropriate regulatory authorities. This foreign regulatory approval process typically includes all of the risks and costs associated with FDA approval described above and may also include additional risks and costs, such as the risk that such foreign regulatory authorities, which often have different regulatory and clinical trial requirements, interpretations and guidance from the FDA, may require additional clinical trials or results for approval of a product candidate, any of which could result in delays, significant additional costs or failure to obtain such regulatory
approval. There can be no assurance, however, that we or our collaborative partners will not have to provide additional information or analysis, or conduct additional clinical trials, before receiving approval to market product candidates.
We have orphan drug designations from the FDA but we may not be able to obtain additional orphan drug designations in the future, or maintain the orphan drug designations or exclusivity for the approved drugs for the treatment of respective indications, or we may be unable to maintain the benefits associated with orphan drug designations, including the potential for market exclusivity.
We have an orphan drug designation in the US for fostamatinib for the treatment of ITP and warm auto immune hemolytic anemia (wAIHA), and for olutasidenib for the treatment of AML. Also, pralsetinib has an orphan drug designation in the US for the treatment of adult patients with metastatic RET fusion-positive NSCLC, for the treatment of advanced or metastatic RET fusion-positive thyroid cancer, and for the treatment of advanced or metastatic RET-mutant medullary thyroid carcinoma. In January 2025, the FDA granted R289 orphan drug designation for the treatment of myelodysplastic syndromes. We may also seek orphan drug designation for other product candidates in the future. Under the Orphan Drug Act, the FDA may grant orphan drug designation to a drug or biologic intended to treat a rare disease or condition, which is defined as one occurring in a patient population of fewer than 200,000 in the US, or a patient population greater than 200,000 in the US where there is no reasonable expectation that the cost of developing the drug will be recovered from sales in the US. In the US, orphan drug designation entitles a party to financial incentives such as opportunities for grant funding towards clinical trial costs, tax advantages and user-fee waivers. In addition, if a product that has orphan drug designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications, including a full NDA, to market the same drug for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity or where the manufacturer is unable to assure sufficient product quantity. At this time, we do not have nor will we seek to apply for orphan drug designation in the EU or the UK in the foreseeable future.
We cannot assure that any future application for orphan drug designation with respect to any other product candidate will be granted. If we are unable to obtain orphan drug designation with respect to other product candidates in the US, we will not be eligible to obtain the period of market exclusivity that could result from orphan drug designation or be afforded the financial incentives associated with orphan drug designation. Even though we have received orphan drug designation for fostamatinib for the treatment of ITP and wAIHA in the US, we may not be the first to obtain marketing approval for the orphan-designated indication due to the uncertainties associated with developing pharmaceutical products or we might not maintain our orphan drug designation. In addition, exclusive marketing rights in the US for fostamatinib for the treatment of ITP, wAIHA or any future product candidate may be limited if we seek approval for an indication broader than the orphan-designated indication or may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition. Further, even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs with different active moieties can be approved for the same condition. Even after an orphan product is approved, the FDA can subsequently approve the same drug with the same active moiety for the same condition if the FDA concludes that the later drug is safer, more effective, or makes a major contribution to patient care. Orphan drug designation neither shortens the development time or regulatory review time of a drug nor gives the drug any advantage in the regulatory review or approval process.
In addition, Congress is considering updates to the orphan drug provisions of the FDCA in response to a recent 11th Circuit decision. Any changes to the orphan drug provisions could change our opportunities for, or likelihood of success in obtaining, orphan drug exclusivity and would materially adversely affect our business, financial condition, results of operations, cash flows and prospects.
Risks Related to Commercialization
Our prospects are highly dependent on our commercial products. To the extent that the commercial success of our products in the US is diminished or is not commercially successful, our business, financial condition and results of operations may be adversely affected, and the price of our common stock may decline.*
We are focusing a significant portion of our activities and resources on our products, and we believe our prospects are highly dependent on, and a significant portion of the value of our company relates to, our ability to sustain successful commercialization of our products in the US. We have also entered into exclusive commercialization agreements with third
parties to commercialize our products outside the US, and we plan to further enter partnership with existing or other third parties to commercialize our products outside the US in the future.
Sustained successful commercialization of our products is subject to many risks and uncertainties, including the impact of a global pandemic on the successful commercialization in the US, as well as the successful commercialization efforts for our products through our collaborative partners. There are numerous examples of unsuccessful product launches and failures to meet high expectations of market potential, including by pharmaceutical companies with more experience and resources than us.
There are many factors that could cause the commercialization of our products to be unsuccessful, including a number of factors that are outside our control. The commercial success of our products depends on the extent to which patients and physicians accept and adopt our products to treat the related diseases. We also do not know how physicians, patients and payors will respond to our future price increases of our products. Physicians may not prescribe our products and patients may be unwilling to use our products if coverage is not provided or reimbursement is inadequate to cover a significant portion of the cost. Our products compete, and may in the future compete, with currently existing therapies, including generic drugs, and products currently under development. Our competitors, particularly large pharmaceutical companies, may deploy more resources to market, sell and distribute their products. If our efforts are not appropriately resourced to adequately promote our products, the commercial potential of our sales may be diminished. Additionally, any negative development for our products in clinical development in additional indications may adversely impact the commercialization and potential of our products. Thus, significant uncertainty remains regarding the commercial potential of our products.
Market acceptance of our products will depend on a number of factors, including:
•the timing of market introduction of the product as well as competitive products;
•the clinical indications for which the product is approved;
•acceptance by physicians, the medical community and patients of the product as a safe and effective treatment;
•potential future impacts, if any, due to the effects of a global pandemic and the global tensions arising from geopolitical conflicts, including the ongoing Russia-Ukraine war and the Hamas-Israel and Iran conflicts, as well as other conflicts in the Middle East;
•the ability to distinguish safety and efficacy from existing, less expensive generic alternative therapies, if any;
•the convenience of prescribing, administrating and initiating patients on the product and the length of time the patient is on the product;
•the potential and perceived value and advantages of the product over alternative treatments;
•the cost of treatment in relation to alternative treatments, including any similar generic treatments;
•pricing and the availability and timing of coverage and adequate reimbursement by third-party payors and government authorities and the process for obtaining or re-obtaining such coverage;
•a positive HTA concluding that the product is cost-effective and the HTA bodies issuing a positive recommendation for the use of the product as a first or second line of treatment for the granted therapeutic indication;
•the prevalence and severity of adverse side effects; and
•the effectiveness of sales and marketing efforts.
If we are unable to sustain anticipated level of sales growth from our products, or if we fail to achieve anticipated product royalties and collaboration milestones, we may need to reduce our operating expenses, access other sources of cash or otherwise modify our business plans, which could have a negative impact on our business, financial condition and
results of operations. From time to time, our net product sales are negatively impacted by the decrease in level of inventories remaining at our distribution channels.
Our product revenues are subject to seasonal variability, including in the first quarter of each year, due to changes in patient out-of-pocket costs and access dynamics. At the beginning of the year, patients enrolled in commercial and government insurance plans may experience deductible and co-payment resets, and Medicare beneficiaries may re-enter cost-sharing phases, which can increase out-of-pocket costs and adversely affect patient demand, initiation of therapy and adherence. In addition, annual re-enrollment processes, prior authorization renewals and coverage re-verification may result in temporary delays or interruptions in patient access to our products. These factors have caused and may continue to cause variability in our revenues between reporting periods, including lower net product sales in the first quarter relative to subsequent quarters.
We depend on a limited number of wholesale distributors for the distribution of our products, and the loss of, or significant disruption at, any of these distributors could adversely affect our business. The loss of any of these distributors, or a material reduction in their purchases, could materially and adversely affect our product sales and results of operations. In addition, consolidation among wholesale distributors could increase their bargaining power and reduce our margins.
We also may not be successful entering into arrangements with third parties to sell and market one or more of our product candidates or may be unable to do so on terms that are favorable to us. We have limited control over such third parties, including development and commercialization of fostamatinib in Kissei, Grifols, Medison and Knight’s territories, and of olutasidenib in Kissei and Dr. Reddy’s territories. As a consequence of our license agreements with our collaboration partners, we rely heavily upon their regulatory, commercial, medical affairs, market access and other expertise and resources for commercialization of our products in their respective territories outside of the US. We cannot control the amount of resources that our partners dedicate to the commercialization of our products, and our ability to generate revenues from the commercialization of our products by our partners depends on their ability to achieve market acceptance of our products in its approved indications in their respective territories.
Furthermore, foreign sales of our products by our partners could be adversely affected by the imposition of governmental controls, political and economic instability, outbreaks of pandemic diseases, trade restrictions or barriers and changes in tariffs and escalating global trade and political tensions. If our collaborators are unable to successfully complete clinical trials, delay commercialization of our products or do not invest the resources necessary to successfully commercialize our products in international territories where they have been approved, this could reduce the amount of revenue we are due to receive under these license agreements, resulting in harm to our business and operations. If we do not establish and maintain sales and marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
Even if we, or any of our collaborative partners, are able to continue to commercialize our products or any product candidate that we, or they, develop, the product may become subject to unfavorable pricing regulations, third-party payor reimbursement practices or labeling restrictions, all of which may vary from country to country and any of which could harm our business.
The commercial success of any product for which we have obtained regulatory approval, or for which we may obtain regulatory approval in the future will depend substantially on the extent to which the costs of our product or product candidates are or will be paid by third-party payors, including government healthcare programs and private health insurers. There is a significant trend in the healthcare industry by public and private payors to contain or reduce their costs, including by taking the following steps, among others: decreasing the portion of costs payors will cover, ceasing to provide full payment for certain products depending on outcomes, and/or not covering certain products at all. If payors implement any of the foregoing with respect to our products, it would have an adverse impact on our revenue and results of operations. If coverage is not available, or reimbursement is limited, we, or any of our collaborative partners, may not be able to successfully commercialize our products or any of our product candidates in some jurisdictions. Even if coverage is provided, the approved reimbursement amount may not be at a rate that covers our costs, including research, development, manufacture, sale and distribution. In the US, no uniform policy of coverage and reimbursement for products exists among third-party payors; therefore, coverage and reimbursement levels for products can differ significantly from payor to payor. As a result, the coverage determination process is often a time consuming and costly process that may require us to provide scientific, clinical or other support for the use of our products to each payor separately, with no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first instance.
There is significant uncertainty related to third-party payor coverage and reimbursement of newly approved drugs. Marketing approvals, pricing and reimbursement for new drug products vary widely from country to country. Some countries require approval of the sale price of a drug before it can be marketed, which could delay market entry (or, if pricing is not approved, we may be unable to sell at all in a country where we have received regulatory approval for a product. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some countries, the proposed pricing for a drug must be approved before it may be lawfully marketed). In addition, authorities in some countries impose additional obligations, such as HTAs, which assess the performance of a drug in comparison with its cost. The outcome of HTA assessments is judged on a national basis and some payors may not reimburse the use of our products or may reduce the rate of reimbursement for our products and as a result, revenue from such products may decrease.
On January 12, 2025, the new HTA Regulation, Regulation No 2021/2282 on Health Technology Assessment (HTA Regulation) started applying to new cancer medicines and advanced therapy medicinal products, and imposes a new procedure for the assessment of the pricing and reimbursement of medicinal products. The HTA Regulation intends to foster cooperation among EU member states in assessing health technologies and provides a procedure for joint clinical assessments of medicinal products at a centralized level. It requires companies applying for products in scope to make relevant submissions for the joint clinical assessment, in line with a number of prespecified criteria. By 2030 it will apply to all medicinal products.
In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. As a result, we, or any of our collaborative partners, might obtain marketing approval for a product in a particular country, but then be subject to price regulations that delay commercial launch of the product, possibly for lengthy time periods, which may negatively impact the revenues we are able to generate from the sale of the product in that country. In particular, we cannot predict to what extent the effects of a global pandemic, depending on its scale and duration, may disrupt global healthcare systems and access to our products or result in a widespread loss of individual health insurance coverage due to unemployment, a shift from commercial payor coverage to government payor coverage, or an increase in demand for patient assistance and/or free drug programs, any of which would adversely affect access to and demand for our products and our net sales. Adverse pricing limitations may also hinder our ability or the ability of any future collaborators to recoup our or their investment in one or more product candidates, even if our product candidates obtain marketing approval. Further, even if favorable coverage and reimbursement status is attained for one or more products for which we or our collaborative partners receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
Patients who are provided medical treatment for their conditions generally rely on third-party payors to reimburse all or part of the costs associated with their treatment. Therefore, our ability, and the ability of any of our collaborative partners, to successfully commercialize our products or any of our product candidates will depend in part on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from third-party payors.
Additionally, the labeling ultimately approved for any of our product candidates for which we have or may obtain regulatory approval may include restrictions on their uses and may be subject to ongoing FDA or international regulatory authority requirements governing the labeling, packaging, storage, distribution, safety surveillance, advertising, promotion, record-keeping and reporting of safety and other post-market information. If we or any of our collaborative partners do not timely obtain or comply with the labeling approval by the FDA or international regulatory authorities on any of our product candidates, it may delay or inhibit our ability to successfully commercialize our products and generate revenues.
If we are unable to successfully market and distribute our products and retain experienced commercial personnel, our business will be substantially harmed.
We continuously expend significant time and resources to maintain a sales force that is credible and compliant with applicable laws in marketing our products. In addition, we must continually train our sales force to ensure that an appropriate and compliant message about our products is being delivered. If we are unable to effectively train our sales force and equip them with compliant and effective materials, including medical and sales literature to help them appropriately inform and educate healthcare providers regarding the potential benefits and proper administration of our products, our efforts to successfully commercialize our products could be put in jeopardy, which would negatively impact our ability to generate product revenues.
We have established our distribution, sales, marketing and market access capabilities, all of which will be necessary to successfully commercialize our products. As a result, we will be required to expend significant time and resources to market, sell, and distribute our products to hematologists and hematologist-oncologists. There is no guarantee that the marketing strategies we have developed, or the distribution, sales, marketing and market access capabilities that we have developed will be successful. Particularly, we are dependent on third-party logistics, specialty pharmacies and distribution partners in the distribution of our products. If they are unable to perform effectively or if they do not provide efficient distribution of the medicine to patients, our business may be harmed.
Maintaining our sales, marketing, market access and product distribution capabilities requires significant resources, and there are numerous risks involved with managing our commercial team, including our potential inability to successfully train, retain and incentivize adequate numbers of qualified and effective sales and marketing personnel. We are also competing for talent with numerous commercial and pre-commercial-stage oncology-focused biotechnology companies seeking to build out their commercial organizations, as well as other large pharmaceutical organizations that have extensive, well-funded and more experienced sales and marketing operations, and we may be unable to maintain or adequately scale our commercial organization as a result of such competition. If we cannot maintain effective sales, marketing, market access, and product distribution capabilities, we may be unable to realize the commercial potential of our products. Also, to the extent that the commercial opportunities for our products grow over time, we may not properly judge the requisite size and experience of our current commercialization teams or the level of distribution necessary to market and sell our products, which could have an adverse impact on our business, financial condition and results of operations.
We may not be able to successfully develop or commercialize our product candidates if problems arise in the clinical testing and approval process.
The activities associated with the research, development and commercialization of our products and other product candidates in our pipeline must undergo extensive clinical trials, which can take many years and require substantial expenditures, subject to extensive regulation by the FDA and other regulatory agencies in the US and by comparable authorities in other countries. The process of obtaining regulatory approvals in the US and other foreign jurisdictions is expensive, and lengthy, if approval is obtained at all.
Our clinical trials may fail to produce results satisfactory to the FDA or regulatory authorities in other jurisdictions. The regulatory process also requires preclinical testing, and data obtained from preclinical and clinical activities are susceptible to varying interpretations. The FDA has substantial discretion in the approval process and may refuse to approve any NDA or sNDA and decide that our data is insufficient for approval and require additional preclinical, clinical or other studies. Varying interpretations of the data obtained from preclinical and clinical testing could delay, limit or prevent regulatory approval of our products for any individual, additional indications. For example, in June 2022, we announced that the top-line results from our Phase 3 trial in wAIHA did not demonstrate statistical significance in the primary efficacy endpoint of durable hemoglobin response in the overall study population. Based on the result of the trial and the guidance from the FDA, we did not file an sNDA for wAIHA.
It is also possible that we could experience delays in the timing of our interactions with regulatory authorities due to absenteeism by governmental employees or the diversion of regulatory authority efforts and attention to approval of other therapeutics, or other public health emergencies including a global pandemic, which could delay or limit our ability to make planned regulatory submissions or develop and commercialize our product candidates on anticipated timelines.
In addition, delays or rejections may be encountered based upon changes in regulatory policy for product approval during the period of product development and regulatory agency review, which may cause delays in the approval or rejection of an application for our products or for our other product candidates.
Commercialization of our product candidates depends upon successful completion of extensive preclinical studies and clinical trials to demonstrate their safety and efficacy for humans. Preclinical testing and clinical development are long, expensive and uncertain processes.
In connection with clinical trials of our product candidates, we may face the following risks among others:
•the product candidate may not prove to be effective;
•the product candidate may cause harmful side effects;
•the clinical results may not replicate the results of earlier, smaller trials;
•we or third parties with whom we collaborate, may be significantly impacted by force majeure events;
•we, or the FDA or similar foreign regulatory authorities, may delay, terminate or suspend the trials;
•our results may not be statistically significant;
•patient recruitment and enrollment may be slower than expected;
•patients may drop out of the trials or otherwise not enroll; and
•regulatory and clinical trial requirements, interpretations or guidance may change.
We do not know whether we will be permitted to undertake clinical trials of potential products beyond the trials already concluded and the trials currently in process. It will take us or our collaborative partners several years to complete any such testing, and failure can occur at any stage of testing. Interim results of trials do not necessarily predict final results, and acceptable results in early trials may not be repeated in later trials. A number of companies in the pharmaceutical industry, including biotechnology companies, have suffered significant setbacks in advanced clinical trials, even after achieving promising results in earlier trials.
Further, evolving FDA standards may cause additional setbacks. Alterations to clinical trial requirements, including due to judicial challenges, may affect recruitment and retention of patients and may hinder or delay a clinical trial. Further, changes to data requirements may cause FDA or comparable foreign regulatory authorities to disagree with data from preclinical studies or clinical trials, and may require further studies. Changes to trial requirements or trial data may increase costs and delay product development.
General Risk Factors
Global economic conditions could adversely impact our business.*
Deterioration in the macroeconomic economy could lead to losses or defaults by our customers or suppliers, which in turn, could have a material adverse effect on our current and/or projected business operations and results of operations and financial condition. The global financial markets and economy are currently, and have from time to time experienced extreme volatility and disruptions, including severely diminished liquidity and credit availability, rising interest and inflation rates, declines in consumer confidence, declines in economic growth, increases in unemployment rates and uncertainty about economic stability.
Any significant deterioration in the US economy would likely affect the operation of our business and ability to raise capital. In addition, concerns regarding the US federal debt ceiling and budget deficit have increased the risk of credit-rating downgrades and economic slowdowns, or a recession in the US. Although US lawmakers passed legislation to raise the federal debt ceiling on multiple occasions, ratings agencies have lowered or threatened to lower the long-term sovereign credit rating on the US. The impact of this or any further downgrades to the US government’s sovereign credit rating or its perceived creditworthiness could adversely affect the US and global financial markets and economic conditions.
The global financial markets and economy may also be adversely affected by the current or anticipated impact of geopolitical conflict, including the ongoing Russian-Ukrainian war, and the Hamas-Israel conflict, which is currently subject to a fragile ceasefire with ongoing hostilities and risk of renewed escalation, as well as the conflict involving Iran and other actors in the Middle East and the potential for those conflicts to escalate even further. The conflict involving Iran, including military actions by the US and Israel and retaliatory responses across the region has contributed to significant volatility in global energy markets, disruptions to key shipping routes such as the Strait of Hormuz, and broader instability in commodity prices and supply chains. These developments have led to increased oil and gas prices, heightened inflationary pressures, and volatility in global equity and debt markets, and may continue to affect global economic growth. Continued or increased disruptions to critical infrastructure and trade routes, as well as the potential for further regional escalation, could exacerbate these impacts and contribute to prolonged economic uncertainty, including risks of stagflation or recession in certain markets. Sanctions imposed by the US and other countries in response to such conflicts, including those targeting Iran, Russia, and other affected regions, may further adversely impact the financial markets and the global
economy. In addition, any retaliatory actions, countermeasures, or expansion of hostilities, whether through direct military engagement, cyber activity, or disruption of global trade and energy supplies, could intensify market volatility and economic instability.
The US government has indicated its intent to alter its approach to international trade policy and in some cases to renegotiate, or potentially terminate, certain existing bilateral or multi-lateral trade agreements and treaties with foreign countries. In addition, the US government has initiated tariffs on certain foreign goods. Related to this action, certain foreign governments have instituted tariffs on certain US goods. Recently imposed US tariffs on patented pharmaceutical products and their ingredients could increase our costs and adversely affect our business. On April 2, 2026, President Trump issued a Proclamation under Section 232 of the Trade Expansion Act of 1962 imposing a 100% ad valorem tariff on imports of patented pharmaceutical products and associated pharmaceutical ingredients. For the 17 large pharmaceutical companies identified in Annex III of the Proclamation, the tariffs become effective on July 31, 2026; for all other importers, including smaller pharmaceutical companies that rely on contract manufacturers, the tariffs become effective on September 29, 2026. The Proclamation provides for reduced tariff rates in certain circumstances. The Proclamation also provides for a 0% tariff rate on certain specialty pharmaceutical products, including drugs for which all approved indications carry orphan drug designation under the Orphan Drug Act, subject to certain conditions, including that the products are from countries that have entered into trade agreements with the US or meet an urgent public health need. Each of our three commercial products has received orphan drug designation from the FDA for its approved indications. However, we cannot provide assurance that our products will qualify for the orphan drug exemption, as the exemption is subject to US government determinations that have not yet been made, and the scope and application of the exemption remain uncertain. We rely on third-party contract manufacturers, certain of which are located outside the United States or source raw materials, including active pharmaceutical ingredients and excipients, from foreign countries that may be subject to tariffs under the Proclamation. To the extent the tariffs apply to our products or their inputs, they could significantly increase our cost of product sales, reduce our gross margins, and adversely affect our results of operations. We may be unable to pass increased costs on to our customers or patients, and we may not be able to identify alternative suppliers or manufacturing arrangements on acceptable terms or in a timely manner. In addition, the tariffs could increase the cost of our research and development activities to the extent we rely on imported materials for clinical supply manufacturing. The Proclamation may also affect our collaboration partners outside the US. Certain of our partners import and commercialize our products in territories that may be subject to retaliatory trade measures, which could adversely affect their ability to commercialize our products and reduce the royalty and milestone revenues we receive under our collaboration agreements. The full impact of the Proclamation on our business will depend on factors including the final determinations regarding orphan drug and other exemptions, the outcome of any legal challenges to the Proclamation, and whether additional tariff actions are taken. It remains unclear what the US Administration or foreign governments will or will not do with respect to tariffs or other international trade agreements and policies. A trade war or further governmental action related to tariffs or international trade agreements or policies has the potential to disrupt our research activities, affect our suppliers and/or the US or global economy or certain sectors thereof and, thus, could adversely impact our businesses.
Shareholder activism and private securities-related litigation could cause material disruption to our business.
Publicly traded companies have increasingly become subject to campaigns by our stakeholders, including investors, and more recently regulatory organizations advocating corporate actions such as actions related to Environmental Social Governance (ESG) matters, impacts of climate change, financial restructuring, increased borrowing, dividends, share repurchases and even sales of assets or the entire company. Responding to proxy contests and other actions by such activist investors or others in the future could be costly and time-consuming, disrupt our operations and divert the attention of our Board of Directors and senior management from the pursuit of our business strategies, which could adversely affect our results of operations and financial condition.
There is increasing focus from investors, regulators and other stakeholders on corporate responsibility, including ESG factors, which could adversely affect our business. Certain investors and advocacy groups use ESG criteria to inform investment and voting decisions, and may choose not to invest in, or take actions against, companies they perceive as having inadequate ESG practices. In addition, third-party ESG ratings are widely used to evaluate companies, and a low or declining rating could negatively impact investor interest in our common stock. Evolving ESG standards and expectations may also require us to incur additional costs to implement new policies, procedures or initiatives. At the same time, there is increasing political and regulatory scrutiny of ESG practices, including actions by the US federal government, state officials and courts that seek to limit or challenge ESG-related initiatives. These developments have contributed to a fragmented and rapidly evolving regulatory and litigation landscape, and may expose us to additional legal, compliance and reputational risks regardless of our approach to ESG. Any of these factors could adversely affect our reputation, investor
base and access to capital, and could materially and adversely affect our business, financial condition and results of operations.
Securities-related class action lawsuits and/or derivative lawsuits have often been brought against companies, including biotechnology and biopharmaceutical companies, that experience volatility in the market price of their securities. It is possible that such lawsuit will be filed, or allegations from stockholders with this matter. Such lawsuits and any other related lawsuits are subject to inherent uncertainties, and the actual defense and disposition costs will depend upon many unknown factors. The outcome of such lawsuits is necessarily uncertain. We could be forced to expend significant resources in the defense of the pending lawsuits and any additional lawsuits, and we may not prevail.
Anti-takeover provisions in our charter documents and under Delaware law may make an acquisition of us, which may be beneficial to our stockholders, more difficult.
Provisions of our Amended and Restated Certificate of Incorporation and our Amended and Restated Bylaws (our Bylaws), as well as provisions of Delaware law, could make it more difficult for a third party to acquire us, even if doing so would benefit our stockholders. These provisions:
•establish that members of our Board of Directors may be removed only for cause upon the affirmative vote of stockholders owning a majority of our capital stock;
•authorize the issuance of “blank check” preferred stock that could be issued by our Board of Directors to increase the number of outstanding shares and thwart a takeover attempt;
•limit who may call a special meeting of stockholders;
•prohibit stockholder action by written consent, thereby requiring all stockholder actions to be taken at a meeting of our stockholders;
•establish advance notice requirements for nominations for election to our Board of Directors or for proposing matters that can be acted upon at stockholder meetings;
•provide for staggered terms for our Board of Directors; and
•provide that the authorized number of directors may be changed only by a resolution of our Board of Directors.
In addition, Section 203 of the Delaware General Corporation Law (DGCL), which imposes certain restrictions relating to transactions with major stockholders, may discourage, delay or prevent a third party from acquiring us.
Our Bylaws designate a state or federal court located within the State of Delaware as the sole and exclusive forum for substantially all disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our current or former directors, officers, stockholders, or other employees.
Our Bylaws provide that, unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware shall be the sole and exclusive forum for (i) any derivative action or proceeding brought on behalf of us under Delaware law, (ii) any action asserting a claim of breach of a fiduciary duty by any current or former director, officer, or other employee of ours that is owed to us or our stockholders, (iii) any action asserting a claim against us or any of our directors, officers, or other employees arising pursuant to any provision of the DGCL or our Amended and Restated Certificate of Incorporation and our Bylaws (as either may be amended from time to time), (iv) any action asserting a claim against us governed by the internal affairs doctrine, or (v) any other action asserting an “internal corporate claim,” as defined under Section 115 of the DGCL. The forgoing provisions do not apply to any claims arising under the Securities Act and, unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States will be the sole and exclusive forum for resolving any action asserting a claim arising under the Securities Act.
These choice of forum provisions may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or any of our current or former directors, officers, or other employees, which may discourage
lawsuits with respect to such claims. There is uncertainty as to whether a court would enforce such provisions, and the enforceability of similar choice of forum provisions in other companies’ charter documents has been challenged in legal proceedings. It is possible that a court could find these types of provisions to be inapplicable or unenforceable, and if a court were to find the choice of forum provision to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions, which could harm our business, results of operations, and financial condition.
Increasing use of social media could give rise to liability and may harm our business.
We and our employees are increasingly utilizing social media tools and our website as a means of communication. Despite our efforts to monitor evolving social media communication guidelines and comply with applicable laws, regulations and national and EU codes of conduct, there is risk that the unauthorized use of social media by us or our employees to communicate about our products or business, sharing of publications in unintended audiences in other jurisdictions, or any inadvertent promotional activity or disclosure of material, nonpublic information through these means, may cause us to be found in violation of applicable laws and regulations, which may give rise to liability and result in harm to our business. In addition, there is also risk of inappropriate disclosure of sensitive information, which could result in significant legal and financial exposure and reputational damages that could potentially have an adverse impact on our business, financial condition and results of operations. Furthermore, negative posts or comments about us or our products on social media could seriously damage our reputation, brand image and goodwill.
Our future success depends on our ability to attract and retain key employees and relationships.
We are highly dependent on the commercial, research and development, clinical, business development, financial and legal expertise of our executive officers, as well as the other principal members of our management. We expect to continue hiring and retaining qualified personnel, which is critical to our success. Replacing key employees and executive officers may be difficult and may take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to successfully develop, gain regulatory approval of and commercialize drugs. Competition to hire from this limited pool is intense, and we may be unable to hire, train, retain or motivate these key personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. In particular, our research programs depend on our ability to attract and retain highly skilled chemists, other scientists, and development, regulatory and clinical personnel. If we lose the services of any of our key personnel, our research and development efforts could be seriously and adversely affected. Our employees can terminate their employment with us at any time.
Our facilities are located near known earthquake fault zones, and the occurrence of an earthquake or other catastrophic disaster could cause damage to our facilities and equipment, which could require us to cease or curtail operations.
Our facilities are located in the San Francisco Bay Area near known earthquake fault zones and are vulnerable to significant damage from earthquakes. We are also vulnerable to damage from other types of disasters, including fires, floods, power loss, communications failures and similar events. If any disaster were to occur, our ability to operate our business at our facilities would be seriously, or potentially completely, impaired, and our research could be lost or destroyed. In addition, the unique nature of our research activities and of much of our equipment could make it difficult for us to recover from a disaster. The insurance we maintain may not be adequate to cover our losses resulting from disasters or other business interruptions.
Item 2. Unregistered Sales of Equity Securities and Use of Proceeds
Unregistered Sales of Equity Securities
There were no sales of unregistered equity securities during the three months ended March 31, 2026.
Issuer Purchases of Equity Securities
The following table summarizes repurchases of our common stock during the three months ended March 31, 2026:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| Period | | Total number of shares purchased(1) | | Average price paid per share | | Total number of shares purchased as part of publicly announced plans or programs | | Maximum approximate dollar value of shares that may yet be purchased under the plans or programs |
| January 1 - January 31 | | — | | | $ | — | | | — | | | — | |
| February 1 - February 28 | | 105,728 | | | $ | 35.37 | | | — | | | — | |
| March 1 - March 31 | | — | | | $ | — | | | — | | | — | |
(1) Represents shares withheld by us to satisfy tax withholding obligations in connection with the vesting of RSUs. These share repurchases were not made pursuant to a publicly announced share repurchase program.
Item 3. Defaults Upon Senior Securities
None.
Item 4. Mine Safety Disclosures
Not applicable.
Item 5. Other Information
Securities Trading Plans of Directors and Executive Officers
During the three months ended March 31, 2026, no directors or executive officers adopted or terminated any contract, instruction or written plan for the purchase or sale of our securities that was intended to satisfy the affirmative defense conditions of Rule 10b5-1(c) or any “non-Rule 10b5-1 trading arrangement” as defined in Item 408 of Regulation S-K under the Securities Exchange.
Item 6. Exhibits
The exhibits listed on the accompanying index to exhibits are filed or incorporated by reference (as stated therein) as part of this Quarterly Report on Form 10-Q.
| | | | | | | | |
Exhibit
Number | | Description of Document |
| | |
| 3.1 | | |
| | |
| 3.2 | | |
| | |
| 3.3 | | |
| | |
| 3.4 | | |
| | |
| 3.5 | | |
| | |
| 4.1 | | |
| | |
| 4.2 | | |
| | |
| 31.1# | | |
| | |
| 31.2# | | |
| | |
| 32.1#* | | |
| | |
| 101.INS | | Inline XBRL Instance Document – the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document. |
| | |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema Document. |
| | |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase Document. |
| | |
| 101.LAB | | Inline XBRL Taxonomy Extension Labels Linkbase Document. |
| | |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase Document. |
| | |
| 101.DEF | | Inline XBRL Taxonomy Extension Definition Linkbase Document. |
| | |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
______________________________________________________________________
#Filed herewith.
*The certifications attached as Exhibit 32.1 accompany this Quarterly Report on Form 10-Q pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, and shall not be deemed “filed” by the registrant for purposes of Section 18 of the Exchange Act.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | |
| RIGEL PHARMACEUTICALS, INC. |
| | |
| By: | /s/ RAUL R. RODRIGUEZ |
| | Raul R. Rodriguez |
| | Chief Executive Officer
(Principal Executive Officer) |
| Date: | May 5, 2026 |
| | |
| | |
| By: | /s/ DEAN L. SCHORNO |
| | Dean L. Schorno |
| | Chief Financial Officer
(Principal Financial Officer) |
| Date: | May 5, 2026 |